WHY FDA APPROVAL MATTERS FOR MEDICAL DEVICE BUYERS: A COMPREHENSIVE GUIDE TO SAFE AND EFFECTIVE HEALTHCARE PROCUREMENT
In the complex world of healthcare procurement, medical device buyers face countless decisions that directly impact patient safety, clinical outcomes, and organizational success. Among all the factors that influence purchasing decisions, FDA approval stands as the most critical indicator of a device’s safety, effectiveness, and regulatory compliance. Understanding why FDA approval matters is essential for healthcare administrators, procurement specialists, and clinical decision-makers who are responsible for selecting medical equipment that will serve patients and providers effectively.

Understanding the FDA Regulatory Framework
The Food and Drug Administration’s Center for Devices and Radiological Health (CDRH) serves as the primary regulatory authority responsible for overseeing medical devices sold in the United States. This comprehensive regulatory framework was established through the Medical Device Amendments of 1976, which granted the FDA authority to regulate medical devices before they enter the marketplace FDA Device Regulation.
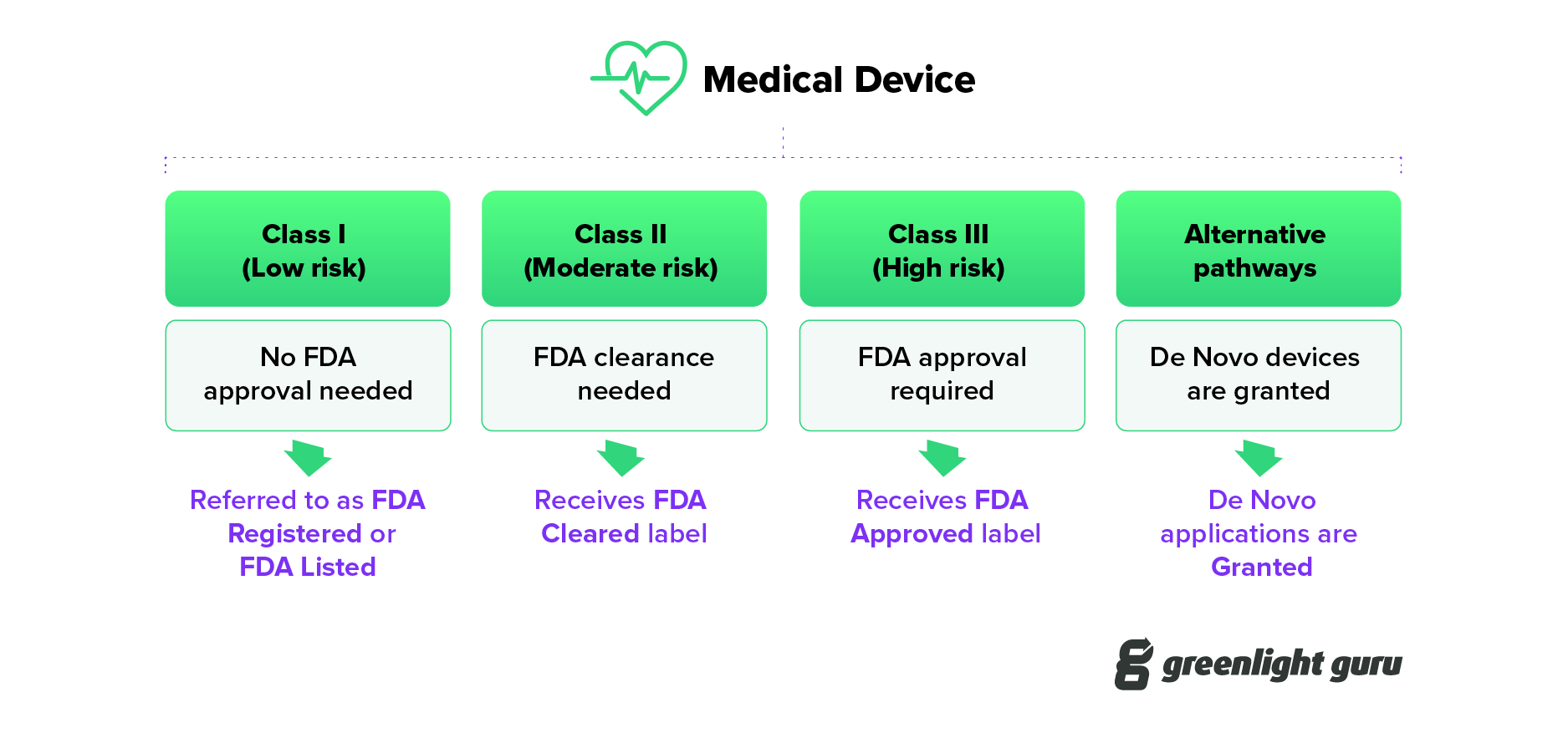
The FDA classifies medical devices into three distinct categories based on their risk profiles and complexity:
Class I Devices represent the lowest risk category, including basic instruments like tongue depressors, bandages, and simple surgical instruments. Most Class I devices are subject to general controls and are exempt from premarket notification requirements, though they must still comply with basic FDA regulations.
Class II Devices encompass moderate-risk products such as infusion pumps, surgical drapes, and diagnostic imaging equipment. These devices typically require 510(k) premarket notification to demonstrate substantial equivalence to legally marketed predicate devices.
Class III Devices include the highest-risk products that are life-sustaining or life-supporting, such as heart valves, pacemakers, and implantable defibrillators. These devices require rigorous Premarket Approval (PMA) with extensive clinical data demonstrating safety and effectiveness.
The Critical Importance of FDA Approval for Patient Safety
FDA approval serves as a fundamental guarantee that medical devices have undergone rigorous testing and evaluation to demonstrate their safety and effectiveness. This regulatory oversight is crucial because it establishes that devices meet stringent standards for materials, design, manufacturing, and performance.
Comprehensive Safety Validation
The FDA approval process requires manufacturers to provide extensive documentation demonstrating that their devices are safe for intended use. This includes biocompatibility testing to ensure materials do not cause adverse reactions when in contact with human tissue, electrical safety verification for powered devices, and mechanical testing to confirm devices can withstand normal use conditions without failure.
Clinical Evidence Requirements
Higher-risk devices must undergo clinical trials to generate evidence of safety and effectiveness in real-world patient populations. These studies are designed according to strict protocols and must demonstrate that the benefits of the device outweigh any potential risks. The FDA reviews this clinical data to ensure that the device performs as intended and does not pose unreasonable risks to patients.
Manufacturing Quality Assurance
FDA approval also requires manufacturers to implement Quality System Regulation (QSR) compliance, ensuring that devices are manufactured under controlled conditions with consistent quality. This includes requirements for design controls, purchasing controls, production and process controls, and corrective and preventive actions.

Regulatory Pathways and Their Significance for Buyers
Understanding the different FDA approval pathways helps buyers assess the level of regulatory scrutiny a device has undergone and make informed purchasing decisions.
510(k) Premarket Notification
The 510(k) pathway is the most common route for Class II devices, requiring manufacturers to demonstrate that their device is substantially equivalent to a legally marketed predicate device. This process typically takes 90-120 days and involves detailed comparison of technological characteristics, performance specifications, and intended use.
What This Means for Buyers: Devices cleared through 510(k) have been compared to existing technologies and found to be substantially equivalent in safety and effectiveness. However, buyers should understand that this pathway does not require clinical trials, so the evidence base may be more limited than for PMA devices.
Premarket Approval (PMA)
The PMA pathway is reserved for Class III devices and requires the most comprehensive regulatory review. Manufacturers must provide extensive preclinical and clinical data demonstrating reasonable assurance of safety and effectiveness for the specific intended use.
What This Means for Buyers: PMA-approved devices have undergone the most rigorous regulatory scrutiny, including clinical trials specifically designed to evaluate the device. This provides buyers with the highest level of confidence in the device’s safety and effectiveness.

De Novo Classification
The De Novo pathway provides a route to market for novel devices that do not have appropriate predicate devices for comparison. This pathway allows the FDA to classify new device types and establish appropriate regulatory controls.
What This Means for Buyers: De Novo devices represent innovative technologies that have been evaluated by the FDA for their specific risk profile. Buyers should carefully review the special controls and labeling requirements established for these devices.
Legal and Compliance Implications
Purchasing non-FDA approved devices or devices used outside their approved indications can expose healthcare organizations to significant legal and regulatory risks.
Liability Considerations
Healthcare facilities that purchase and use non-approved devices may face increased liability exposure in the event of device-related injuries or adverse outcomes. FDA approval provides a level of legal protection by demonstrating that due diligence was exercised in device selection.
Malpractice Risk Mitigation
Using FDA-approved devices according to their approved indications and labeling provides a strong defense against malpractice claims. Courts and juries generally view the use of FDA-approved devices as meeting the standard of care, while the use of non-approved devices may be viewed as substandard care.
Regulatory Compliance
Healthcare facilities are required to comply with various regulations that reference FDA approval status. For example, Medicare and Medicaid reimbursement may be contingent upon the use of FDA-approved devices for certain procedures.
Quality Assurance and Performance Standards
FDA approval ensures that devices meet rigorous quality standards throughout their lifecycle, from design and development through manufacturing and post-market surveillance.
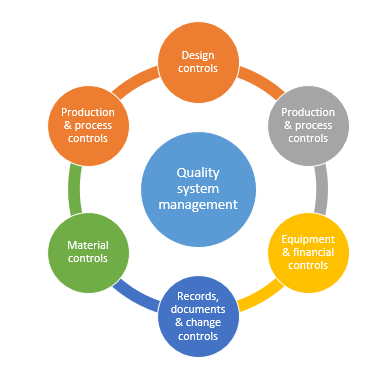
Design Controls
The FDA requires manufacturers to implement design controls that ensure devices are designed to meet user needs and intended uses. This includes design planning, design inputs and outputs, design review, design verification and validation, design transfer, and design changes.
Impact on Device Performance
These design controls ensure that devices are engineered to perform reliably under intended use conditions. For buyers, this translates to more predictable device performance and reduced risk of unexpected failures.
Manufacturing Controls
FDA-approved devices must be manufactured under Current Good Manufacturing Practice (CGMP) requirements, which ensure consistent product quality and safety. This includes requirements for:
- Document controls and record keeping
- Management responsibility and quality system procedures
- Personnel training and qualification
- Facility and equipment controls
- Purchasing and supplier controls
- Production and process controls
- Corrective and preventive actions

Post-Market Surveillance and Ongoing Safety Monitoring
FDA approval is not a one-time event but establishes an ongoing relationship of surveillance and monitoring to ensure continued device safety and effectiveness.
Medical Device Reporting (MDR)
Manufacturers of FDA-approved devices are required to report certain device-related adverse events and malfunctions to the FDA. This post-market surveillance system helps identify safety issues that may not have been apparent during premarket evaluation.
Benefits for Buyers
This ongoing monitoring provides buyers with confidence that safety issues will be identified and addressed promptly. The FDA’s ability to issue safety communications, require labeling changes, or order recalls provides an additional layer of protection for healthcare facilities and patients.
Post-Market Study Requirements
The FDA may require manufacturers to conduct post-market studies to address questions about device safety or effectiveness that arise after approval. These studies provide additional data to inform ongoing use of the device.
Economic Considerations and Value Proposition
While FDA-approved devices may have higher upfront costs compared to non-approved alternatives, the total cost of ownership typically favors approved devices due to several factors.
Reduced Liability Costs
The liability protection provided by FDA approval can result in lower malpractice insurance premiums and reduced exposure to litigation costs. Healthcare facilities that consistently use FDA-approved devices may be viewed more favorably by insurance carriers.
Operational Efficiency
FDA-approved devices are more likely to perform reliably and consistently, reducing downtime, maintenance costs, and the need for device replacement. This operational reliability translates to cost savings over the device lifecycle.
Reimbursement Advantages
Many insurance providers and government payers require the use of FDA-approved devices for reimbursement eligibility. Using non-approved devices may result in claim denials and financial losses for healthcare facilities.

Integration with Healthcare Systems and Interoperability
FDA approval often correlates with better integration capabilities and interoperability with existing healthcare systems and infrastructure.
Standardization Benefits
FDA-approved devices are more likely to comply with industry standards for communication protocols, data formats, and interface specifications. This standardization facilitates integration with electronic health records, medical device networks, and healthcare information systems.
Vendor Support and Longevity
Manufacturers of FDA-approved devices typically provide more robust customer support, training programs, and long-term service commitments. The regulatory investment required for FDA approval creates a stronger business commitment to ongoing product support.
Due Diligence in Device Selection
Healthcare buyers should implement systematic approaches to verify FDA approval status and understand the implications for their specific use cases.
Verification Procedures
Buyers should verify FDA approval status through official FDA databases rather than relying solely on manufacturer claims. The FDA maintains several searchable databases including:
- FDA Device Classification Database
- 510(k) Premarket Notification Database
- PMA Database
- FDA Device Recalls Database
Documentation Requirements
Proper documentation of FDA approval status should be maintained as part of the procurement record. This documentation serves as evidence of due diligence and regulatory compliance for accreditation bodies, auditors, and legal proceedings.

Special Considerations for Emerging Technologies
As healthcare technology evolves rapidly, buyers must understand how FDA approval applies to emerging device categories and innovative technologies.
Software as a Medical Device (SaMD)
The FDA has developed specific guidance for software-based medical devices, including mobile medical applications and artificial intelligence-enabled devices. Buyers should verify that software devices have appropriate FDA clearance or approval for their intended use.
Combination Products
Devices that combine multiple regulated components (device/drug, device/biologic, or device/drug/biologic) may require review by multiple FDA centers. Buyers should understand the regulatory pathway and approval status for all components of combination products.
Best Practices for Medical Device Procurement
Successful medical device procurement requires a systematic approach that prioritizes FDA approval status while considering other important factors.
Multidisciplinary Evaluation Teams
Procurement decisions should involve multidisciplinary teams including clinical users, biomedical engineers, quality assurance personnel, and procurement specialists. This collaborative approach ensures that all relevant factors, including regulatory status, are properly evaluated.
Vendor Assessment
Thorough vendor assessment should include evaluation of:
- FDA registration and listing status
- Quality system certifications
- Post-market surveillance records
- Customer support capabilities
- Financial stability and long-term viability
Risk Management and Mitigation Strategies
Healthcare organizations should implement comprehensive risk management strategies that account for regulatory compliance and device safety considerations.
Risk Assessment Frameworks
Systematic risk assessment should evaluate potential consequences of device failures, including patient safety risks, operational disruptions, and regulatory compliance issues. FDA approval status should be weighted heavily in these risk assessments.
Contingency Planning
Organizations should develop contingency plans for device recalls, safety alerts, and regulatory changes that may affect device availability or use. Having alternative FDA-approved devices identified can help minimize disruptions to patient care.
Global Considerations and International Standards
While this article focuses on FDA approval, buyers should also consider international regulatory standards and approvals, particularly for devices used in global healthcare organizations.
Harmonization Efforts
International harmonization efforts, such as the Medical Device Single Audit Program (MDSAP), are creating more consistent regulatory standards across different countries. However, FDA approval remains the gold standard for devices used in the United States.
CE Marking and Other International Approvals
While CE marking (European Conformity) and other international approvals demonstrate compliance with regulatory standards in their respective markets, they may not be equivalent to FDA approval for use in U.S. healthcare facilities.
Future Trends and Implications
The regulatory landscape for medical devices continues to evolve, with implications for procurement decisions and FDA approval processes.
Digital Health and AI Integration
The increasing integration of digital health technologies and artificial intelligence in medical devices is creating new regulatory challenges and opportunities. The FDA is developing new frameworks for evaluating and approving these technologies.
Real-World Evidence
The FDA is increasingly incorporating real-world evidence into regulatory decision-making, potentially affecting post-market requirements and ongoing approval status for medical devices.

Conclusion: The Imperative of FDA Approval
FDA approval represents far more than regulatory compliance—it serves as a cornerstone of patient safety, quality assurance, and risk management in healthcare procurement. For medical device buyers, understanding and prioritizing FDA approval status is essential for making informed decisions that protect patients, healthcare providers, and organizations.
The comprehensive evaluation process required for FDA approval ensures that devices meet rigorous standards for safety, effectiveness, and quality. This regulatory oversight provides buyers with confidence that approved devices have undergone appropriate testing and validation for their intended use. Moreover, the ongoing post-market surveillance requirements associated with FDA approval create a system of continuous monitoring and improvement that benefits all stakeholders in the healthcare system.
Healthcare organizations that prioritize FDA-approved devices in their procurement decisions demonstrate commitment to patient safety, regulatory compliance, and quality care. While the initial investment in FDA-approved devices may be higher, the total cost of ownership typically favors approved devices due to reduced liability exposure, improved reliability, and better integration with healthcare systems.
As the medical device landscape continues to evolve with emerging technologies and changing regulatory requirements, the importance of FDA approval as a benchmark for device selection will only increase. Healthcare buyers who understand and leverage this regulatory framework will be better positioned to make procurement decisions that support their organizations’ missions of providing safe, effective, and high-quality patient care.
The investment in FDA-approved medical devices represents an investment in patient safety, regulatory compliance, and organizational excellence. For healthcare buyers, there is no substitute for the assurance provided by FDA approval in today’s complex and highly regulated healthcare environment.
