COMPREHENSIVE MEDICAL DEVICES DOCUMENTATION
ADVANCED HEALTHCARE TECHNOLOGY: AI-ENABLED AND USB-CONNECTED DIAGNOSTIC EQUIPMENT
EXECUTIVE SUMMARY
The medical device industry is experiencing unprecedented transformation through the integration of Artificial Intelligence (AI) and Universal Serial Bus (USB) connectivity technologies. This comprehensive documentation presents a detailed analysis of 45 cutting-edge medical devices across three critical categories: AI-enabled systems, USB-connected equipment, and hybrid AI+USB solutions.
Modern healthcare demands rapid, accurate, and accessible diagnostic capabilities. AI integration enables predictive analytics, automated pattern recognition, and enhanced diagnostic accuracy, while USB connectivity ensures seamless data management, real-time monitoring, and interoperability with electronic health records (EHR) systems.
The convergence of these technologies represents the future of point-of-care diagnostics, enabling healthcare professionals to deliver precision medicine with improved patient outcomes and operational efficiency.
SECTION 1: AI-ENABLED MEDICAL DEVICES
This section presents 15 state-of-the-art medical devices featuring integrated artificial intelligence capabilities for enhanced diagnostic accuracy, automated analysis, and clinical decision support.
1. Butterfly iQ3 Ultrasound System

| Specification | Details |
|---|---|
| Manufacturer | Butterfly Network, Inc. |
| Device Classification | FDA Class II Medical Device |
| AI Technology | Deep learning algorithms for automated image optimization |
| Connectivity | Wireless, Cloud-based data management |
| Probe Technology | Butterfly on a Chip (BoC) semiconductor technology |
| Display | iOS/Android mobile app interface |
Medical Conditions Detected:
- Cardiovascular: Heart valve abnormalities, pericardial effusion, ventricular function assessment
- Respiratory: Pneumothorax, pleural effusion, lung consolidation
- Abdominal: Gallbladder pathology, kidney stones, abdominal fluid collection
- Obstetric: Fetal development monitoring, pregnancy complications
- Vascular: Deep vein thrombosis, arterial stenosis
2. Eko CORE 500 Digital Stethoscope

| Specification | Details |
|---|---|
| Manufacturer | Eko Health |
| Device Classification | FDA Class II Medical Device |
| AI Capabilities | AI-powered heart murmur detection, ECG analysis |
| Amplification | 40x amplification at peak frequency |
| ECG Leads | 3-lead ECG capability |
| Battery Life | Up to 9 hours continuous use |
Medical Conditions Detected:
- Cardiovascular: Heart murmurs, atrial fibrillation, heart valve disease
- Arrhythmias: Atrial fibrillation detection with 96% accuracy
- Structural Heart Disease: Valvular abnormalities, cardiomyopathy indicators
- Heart Failure: Early detection of cardiac dysfunction
3. PMcardio AI ECG Analysis

| Specification | Details |
|---|---|
| Manufacturer | Powerful Medical |
| Device Classification | CE Marked, FDA De Novo pathway |
| AI Algorithm | Deep neural networks for ECG interpretation |
| Analysis Speed | Real-time interpretation within seconds |
| Supported Leads | 12-lead, 15-lead, 18-lead ECG systems |
| Integration | Cloud-based API, mobile app |
Medical Conditions Detected:
- Acute Coronary Syndromes: STEMI, NSTEMI, unstable angina
- Arrhythmias: Atrial fibrillation, ventricular tachycardia, heart blocks
- Structural Abnormalities: Left ventricular hypertrophy, chamber enlargement
- Electrolyte Imbalances: Hyperkalemia, hypokalemia effects
- Conduction Disorders: Bundle branch blocks, AV blocks
4. GE Vscan Air SL with Caption AI

| Specification | Details |
|---|---|
| Manufacturer | GE HealthCare |
| Device Classification | FDA 510(k) Cleared |
| AI Technology | Caption AI for automated cardiac measurements |
| Probe Type | Phased array cardiac probe |
| Display | 5.1″ touchscreen display |
| Connectivity | Wi-Fi, Bluetooth, USB-C |
Medical Conditions Detected:
- Cardiac Function: Ejection fraction assessment, wall motion abnormalities
- Valvular Disease: Mitral regurgitation, aortic stenosis
- Heart Failure: Systolic and diastolic dysfunction
- Pericardial Disease: Pericardial effusion, tamponade
- Vascular Assessment: Carotid artery evaluation, DVT screening
5. Philips Lumify Ultrasound
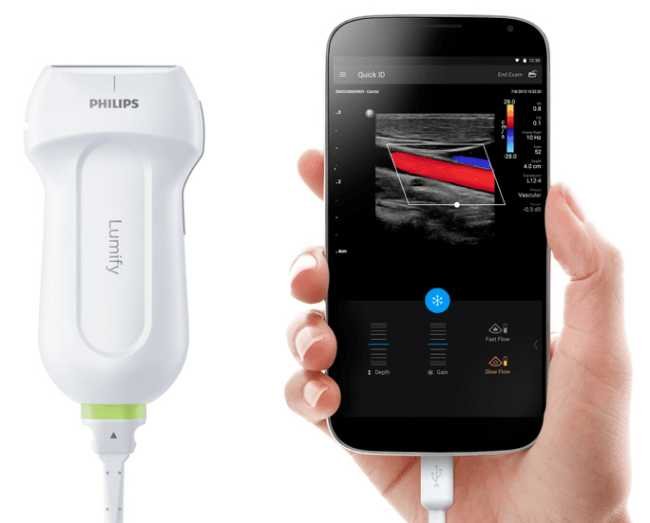
| Specification | Details |
|---|---|
| Manufacturer | Philips Healthcare |
| Device Classification | FDA 510(k) Cleared |
| AI Features | Auto Doppler, Smart Exam protocols |
| Probe Options | Linear (L12-4), Curved (C5-2), Phased (S4-1) |
| Platform | iOS and Android compatible |
| Cloud Storage | Philips HealthSuite integrated |
Medical Conditions Detected:
- Point-of-Care: FAST exam, cardiac assessment, lung pathology
- Emergency Medicine: Pneumothorax, abdominal trauma, shock evaluation
- Critical Care: Fluid status assessment, ventilator management
- Primary Care: Thyroid nodules, gallbladder disease, pregnancy monitoring
- MSK Applications: Joint effusions, soft tissue masses
6. Clarius AI Ultrasound Scanners
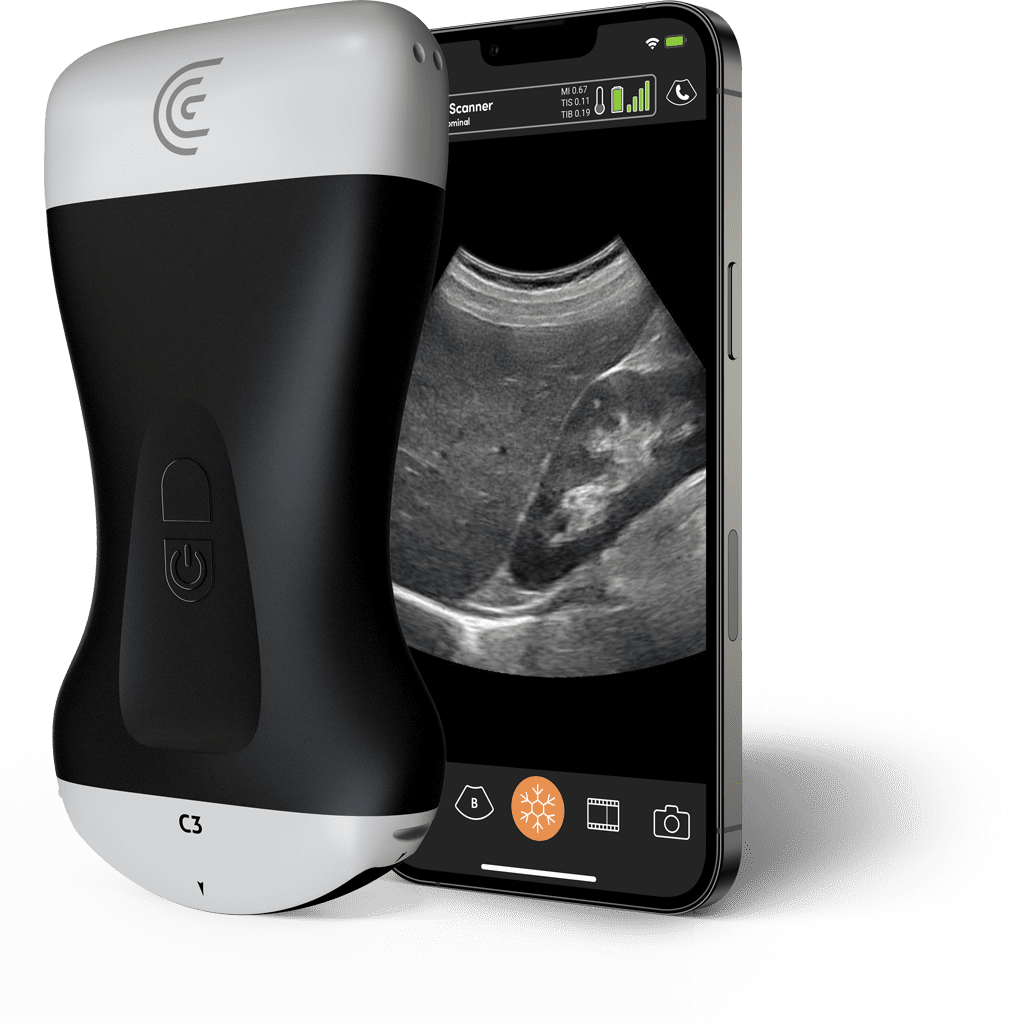
| Specification | Details |
|---|---|
| Manufacturer | Clarius Mobile Health |
| Device Classification | FDA 510(k) Cleared, Health Canada Licensed |
| AI Technology | Clarius AI for automated measurements and needle guidance |
| Scanner Types | C3, L15, L20, EC7 HD3 models |
| Imaging Depth | Up to 30 cm (model dependent) |
| Battery Life | Up to 2 hours continuous scanning |
Medical Conditions Detected:
- MSK Conditions: Carpal tunnel syndrome (FDA-cleared AI), tendon injuries
- Abdominal Pathology: Gallstones, kidney stones, abdominal aortic aneurysm
- Cardiac Assessment: Pericardial effusion, basic cardiac function
- Vascular Conditions: DVT screening, arterial stenosis
- Bladder Assessment: Urinary retention, bladder volume measurement
7. Mindray TE7 Max Ultrasound
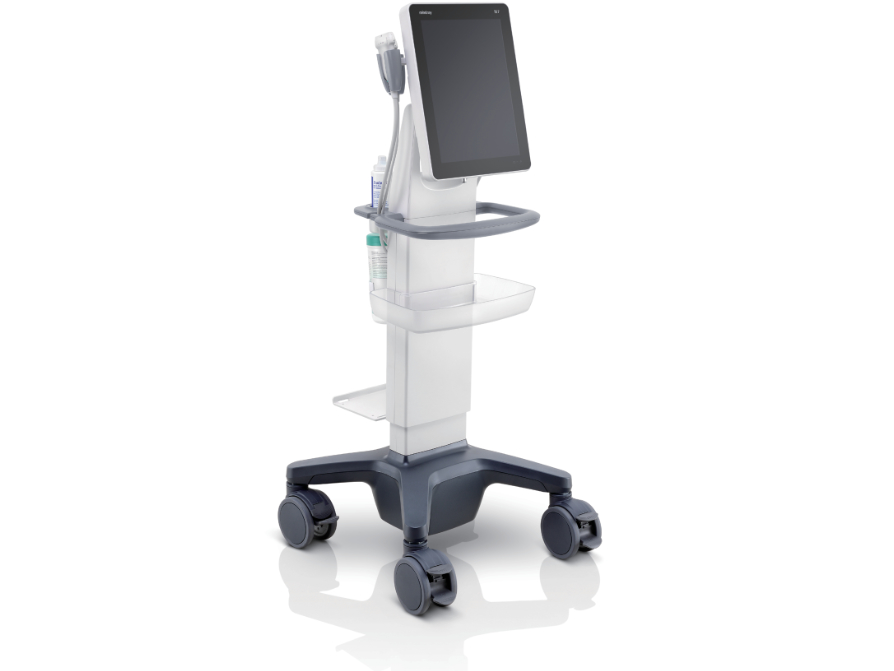
| Specification | Details |
|---|---|
| Manufacturer | Mindray Medical |
| Device Classification | FDA 510(k) Cleared, CE Marked |
| AI Features | Smart IMT, Auto EF, Smart NT |
| Display | 15.6″ LED monitor with touch panel |
| Probe Connectors | 4 active probe connectors |
| Advanced Features | 4D imaging, contrast imaging, elastography |
Medical Conditions Detected:
- Cardiovascular: Coronary artery disease risk assessment, cardiac function
- Obstetrics: Fetal anomalies, growth restriction, nuchal translucency
- Abdominal: Liver pathology, pancreatic disorders, renal disease
- Vascular: Carotid intima-media thickness, arterial stenosis
- Emergency: Trauma assessment, shock evaluation, critical care monitoring
8. Zebra Medical Vision AI

| Specification | Details |
|---|---|
| Manufacturer | Zebra Medical Vision (Nanox subsidiary) |
| Device Classification | FDA 510(k) Cleared multiple algorithms |
| AI Technology | Deep learning for medical imaging analysis |
| Imaging Modalities | CT, X-ray, MRI compatible |
| Integration | PACS integration, cloud-based deployment |
| Processing Speed | Real-time analysis and reporting |
Medical Conditions Detected:
- Chest Imaging: Pneumothorax, pleural effusion, lung nodules
- Cardiac: Coronary artery calcium scoring, cardiac dysfunction
- Bone Health: Osteoporosis, compression fractures
- Liver Disease: Fatty liver, liver lesions
- Brain Imaging: Intracranial hemorrhage, brain atrophy
9. Withings BeamO 4-in-1 Health Scanner

| Specification | Details |
|---|---|
| Manufacturer | Withings |
| Device Classification | FDA 510(k) Pending |
| AI Features | Smart fever detection, automated health assessment |
| Functions | Thermometer, stethoscope, pulse oximeter, otoscope |
| Connectivity | Wi-Fi, Bluetooth, Health Mate app integration |
| Power | Rechargeable battery, USB-C charging |
Medical Conditions Detected:
- Infectious Diseases: Fever detection, early illness screening
- Respiratory: Heart and lung sound abnormalities
- Cardiovascular: Heart murmurs, arrhythmias
- ENT Conditions: Ear infections, tympanic membrane abnormalities
- Oxygen Saturation: Hypoxemia, respiratory compromise
10. EchoNous Kosmos AI POCUS
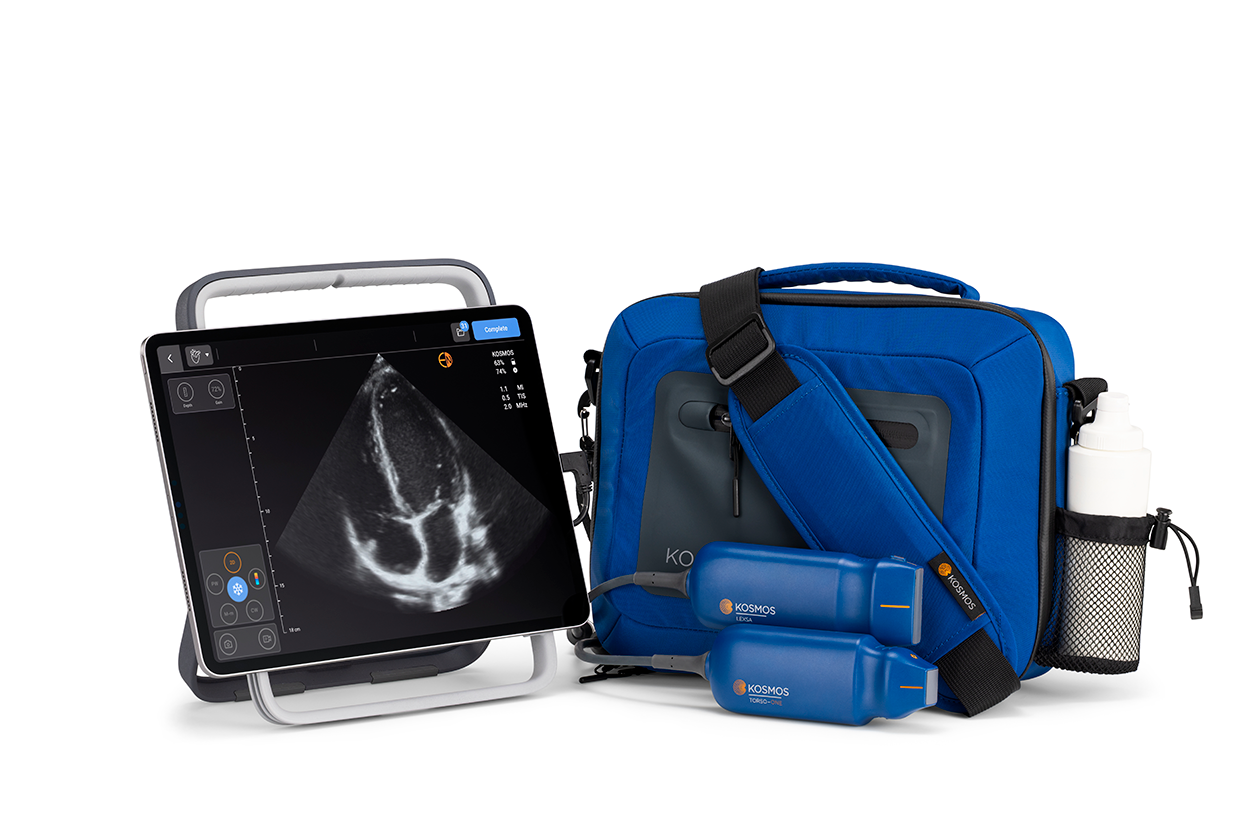
| Specification | Details |
|---|---|
| Manufacturer | EchoNous, Inc. |
| Device Classification | FDA 510(k) Cleared |
| AI Technology | AI-guided imaging protocols and measurements |
| Probe Types | Torso-One multi-frequency probe |
| Display | Tablet-based interface |
| Portability | Compact design for point-of-care use |
Medical Conditions Detected:
- Critical Care: Shock evaluation, fluid status assessment
- Emergency Medicine: FAST exam, cardiac arrest protocols
- Pulmonary: Pneumonia, pulmonary edema, pleural effusion
- Cardiac: Pericardial effusion, basic systolic function
- Abdominal: Free fluid detection, bladder assessment
11. Exo Iris AI Ultrasound

| Specification | Details |
|---|---|
| Manufacturer | Exo Imaging, Inc. |
| Device Classification | FDA 510(k) Cleared |
| AI Capabilities | FDA-cleared AI for cardiac and lung diagnosis |
| Design | Handheld, all-in-one device |
| Imaging Technology | Proprietary transducer technology |
| Power | Built-in rechargeable battery |
Medical Conditions Detected:
- Cardiac Pathology: Heart failure, pericardial effusion, valve disease
- Pulmonary Disease: Pneumonia, pneumothorax, pleural effusion
- Critical Care: Hemodynamic assessment, shock evaluation
- Emergency Medicine: Trauma assessment, cardiac arrest
- Primary Care: Basic cardiac screening, lung pathology detection
12. 3M Littmann CORE Digital Stethoscope

| Specification | Details |
|---|---|
| Manufacturer | 3M Health Care |
| Device Classification | FDA 510(k) Cleared |
| AI Integration | Compatible with Eko AI analysis platform |
| Amplification | Up to 40x amplification |
| Connectivity | Bluetooth, mobile app integration |
| Recording | Audio recording and playback capability |
Medical Conditions Detected:
- Cardiac Auscultation: Heart murmurs, gallops, friction rubs
- Pulmonary Assessment: Rales, rhonchi, wheezes, pleural friction rubs
- Vascular Sounds: Bruits, arterial stenosis
- Abdominal Sounds: Bowel sounds, vascular bruits
- General Assessment: Vital signs monitoring, clinical examination
13. AioCare AI Spirometer

| Specification | Details |
|---|---|
| Manufacturer | AioCare (Vyaire Medical) |
| Device Classification | CE Marked, FDA 510(k) Cleared |
| AI Features | Automated cough detection, quality control |
| Connectivity | Bluetooth smartphone connection |
| Measurements | FEV1, FVC, PEF, and other lung function parameters |
| Platform | iOS and Android mobile apps |
Medical Conditions Detected:
- Obstructive Diseases: Asthma, COPD, bronchitis
- Restrictive Diseases: Pulmonary fibrosis, chest wall deformities
- Mixed Patterns: Combined obstructive-restrictive disease
- Disease Monitoring: Treatment response, disease progression
- Occupational Health: Work-related lung disease screening
14. Aluna Digital Spirometer

| Specification | Details |
|---|---|
| Manufacturer | Sparo Labs |
| Device Classification | FDA 510(k) Cleared |
| AI Technology | Machine learning for lung health assessment |
| Connectivity | Bluetooth 5.0, mobile app integration |
| Power | USB-C rechargeable battery |
| Cleaning | Detachable, dishwasher-safe components |
Medical Conditions Detected:
- Respiratory Infections: COVID-19 monitoring, pneumonia recovery
- Chronic Conditions: Asthma control assessment, COPD monitoring
- Lung Capacity: Post-surgical recovery, athletic performance
- Early Detection: Subclinical lung function decline
- Treatment Monitoring: Medication effectiveness, rehabilitation progress
15. Epic Imaging AI Ultrasound
Epic Imaging AI System
| Specification | Details |
|---|---|
| Manufacturer | Epic Imaging |
| Device Classification | FDA 510(k) Pending |
| AI Capabilities | Real-time AI imaging enhancement and analysis |
| Technology | Advanced POCUS with machine learning |
| Processing | Edge computing for real-time analysis |
| Integration | EMR compatible, cloud-based storage |
Medical Conditions Detected:
- Multi-Organ Assessment: Comprehensive point-of-care evaluation
- Critical Care: Hemodynamic monitoring, shock assessment
- Emergency Medicine: Trauma evaluation, rapid diagnosis
- Cardiac Function: Systolic function, valve assessment
- Pulmonary Pathology: Lung sliding, consolidation, effusion
SECTION 2: USB-CONNECTED MEDICAL DEVICES
This section presents 15 medical devices featuring USB connectivity for seamless data transfer, real-time monitoring, and integration with electronic health record systems.
1. Wellue O2Ring USB Pulse Oximeter

| Specification | Details |
|---|---|
| Manufacturer | Wellue Health |
| Device Classification | FDA 510(k) Cleared |
| USB Features | USB-C charging, data synchronization |
| Monitoring | Continuous SpO2 and heart rate tracking |
| Battery Life | 16 hours continuous monitoring |
| Data Storage | 4 sessions of 10 hours each |
Medical Conditions Detected:
- Sleep Disorders: Sleep apnea, hypoxemia during sleep
- Respiratory Conditions: COPD monitoring, oxygen therapy assessment
- Cardiovascular: Heart rate variability, exercise tolerance
- Chronic Monitoring: Long-term oxygen saturation trends
- Post-Operative Care: Recovery monitoring, surgical complications
2. Nonin 3231 USB Pulse Oximeter

| Specification | Details |
|---|---|
| Manufacturer | Nonin Medical, Inc. |
| Device Classification | FDA 510(k) Cleared |
| USB Connectivity | Direct USB connection to PC |
| Accuracy | ±2% SpO2, ±3 BPM pulse rate |
| Power | USB powered, no batteries required |
| Software | nSoftware data management platform |
Medical Conditions Detected:
- Hypoxemia: Low blood oxygen levels detection
- Respiratory Compromise: Acute and chronic respiratory conditions
- Cardiac Monitoring: Heart rate abnormalities, arrhythmias
- Exercise Testing: Fitness assessment, cardiac rehabilitation
- Clinical Research: Accurate data collection for studies
3. iHealth Gluco+ USB Blood Glucose Monitor

| Specification | Details |
|---|---|
| Manufacturer | iHealth Labs |
| Device Classification | FDA 510(k) Cleared |
| USB Features | USB-C charging, data synchronization |
| Connectivity | Bluetooth, Wi-Fi, USB data transfer |
| Memory | 500 test results with time/date stamps |
| Test Time | 5 seconds |
Medical Conditions Detected:
- Diabetes Mellitus: Type 1 and Type 2 diabetes monitoring
- Prediabetes: Glucose intolerance screening
- Hypoglycemia: Low blood sugar detection and alerts
- Hyperglycemia: High blood sugar management
- Gestational Diabetes: Pregnancy-related glucose monitoring
4. MIR Spirobank II Smart USB Spirometer

| Specification | Details |
|---|---|
| Manufacturer | MIR – Medical International Research |
| Device Classification | FDA 510(k) Cleared, CE Marked |
| USB Connectivity | USB connection for PC-based testing |
| Bluetooth | Wireless connectivity to mobile devices |
| Tests | Spirometry, oximetry (optional) |
| Software | WinspiroPRO, MIR Spiro app |
Medical Conditions Detected:
- Obstructive Lung Disease: Asthma, COPD, chronic bronchitis
- Restrictive Lung Disease: Pulmonary fibrosis, neuromuscular disorders
- Mixed Patterns: Combined restrictive-obstructive conditions
- Occupational Screening: Work-related respiratory conditions
- Pre-Operative Assessment: Surgical risk evaluation
5. OMRON Complete BP Monitor with USB

| Specification | Details |
|---|---|
| Manufacturer | OMRON Healthcare |
| Device Classification | FDA 510(k) Cleared |
| USB Features | USB-C charging, data synchronization |
| Measurements | Blood pressure, ECG, AFib detection |
| Connectivity | Bluetooth, Wi-Fi, USB data transfer |
| Memory | 200+ readings with timestamps |
Medical Conditions Detected:
- Hypertension: High blood pressure detection and monitoring
- Hypotension: Low blood pressure identification
- Atrial Fibrillation: Irregular heart rhythm detection
- Cardiovascular Risk: Heart disease risk assessment
- White Coat Syndrome: Stress-related blood pressure elevation
6. USB Digital Thermometer Systems
Professional Series
| Specification | Details |
|---|---|
| Manufacturer | Various (ThermoWorks, Extech, others) |
| Device Classification | FDA 510(k) Cleared (selected models) |
| USB Connectivity | Real-time data logging via USB |
| Accuracy | ±0.1°C (±0.2°F) |
| Response Time | 1-3 seconds |
| Software | Temperature logging and analysis software |
Medical Conditions Detected:
- Fever: Hyperthermia detection and monitoring
- Hypothermia: Low body temperature identification
- Infectious Diseases: Early fever detection for illness screening
- Post-Operative Monitoring: Temperature stability assessment
- Pediatric Care: Accurate temperature measurement in children
7. USB-Connected ECG Machines
12-Lead Configuration
| Specification | Details |
|---|---|
| Manufacturer | Welch Allyn, Mortara, EDAN |
| Device Classification | FDA 510(k) Cleared |
| USB Features | Direct PC connection, data transfer |
| Leads | 12-lead, 15-lead, 18-lead configurations |
| Software | ECG analysis and interpretation software |
| Printing | USB-connected thermal printer support |
Medical Conditions Detected:
- Myocardial Infarction: STEMI, NSTEMI detection
- Arrhythmias: Atrial fibrillation, ventricular tachycardia
- Conduction Disorders: Heart blocks, bundle branch blocks
- Structural Heart Disease: Left ventricular hypertrophy
- Electrolyte Abnormalities: Hyperkalemia, hypokalemia
8. USB Pulse Oximeter Professional Series
Pulse Oximeter
| Specification | Details |
|---|---|
| Manufacturer | Masimo, Nellcor, Covidien |
| Device Classification | FDA 510(k) Cleared |
| USB Connectivity | Real-time data streaming |
| Accuracy | ±2% SpO2, ±3 BPM |
| Monitoring | Continuous or spot-check measurements |
| Power | USB powered or battery backup |
Medical Conditions Detected:
- Hypoxemia: Oxygen desaturation detection
- Sleep Apnea: Overnight oxygen monitoring
- COPD Monitoring: Chronic respiratory disease management
- Exercise Testing: Cardiac stress test monitoring
- Anesthesia Monitoring: Perioperative oxygen saturation
9. USB Blood Pressure Monitors
Monitoring System
| Specification | Details |
|---|---|
| Manufacturer | A&D Medical, Welch Allyn, OMRON |
| Device Classification | FDA 510(k) Cleared |
| USB Features | Direct data download and analysis |
| Cuff Sizes | Adult, large adult, pediatric options |
| Memory | 500+ readings with time stamps |
| Software | Blood pressure management software |
Medical Conditions Detected:
- Hypertension: Stage 1 and Stage 2 high blood pressure
- Hypotension: Low blood pressure conditions
- Orthostatic Changes: Postural blood pressure variations
- Medication Monitoring: Antihypertensive drug effectiveness
- Ambulatory Monitoring: 24-hour blood pressure assessment
10. USB Spirometry Systems
Spirometry System
| Specification | Details |
|---|---|
| Manufacturer | ndd Medical, COSMED, CareFusion |
| Device Classification | FDA 510(k) Cleared |
| USB Connectivity | Real-time data transmission to PC |
| Tests | FVC, FEV1, PEF, MVV, and more |
| Quality Control | ATS/ERS compliance |
| Software | Comprehensive spirometry analysis |
Medical Conditions Detected:
- Asthma: Bronchial hyperresponsiveness, obstruction patterns
- COPD: Chronic obstructive pulmonary disease staging
- Restrictive Disease: Pulmonary fibrosis, chest wall disorders
- Occupational Lung Disease: Work-related respiratory conditions
- Pre-Operative Assessment: Surgical risk stratification
11. USB Glucose Monitoring Kits
Monitoring Kit
| Specification | Details |
|---|---|
| Manufacturer | Abbott, Roche, LifeScan |
| Device Classification | FDA 510(k) Cleared |
| USB Features | Data download and diabetes management |
| Test Time | 5-15 seconds |
| Memory | 250-1000 test results |
| Software | Diabetes management and trend analysis |
Medical Conditions Detected:
- Type 1 Diabetes: Insulin-dependent diabetes management
- Type 2 Diabetes: Non-insulin dependent diabetes monitoring
- Prediabetes: Impaired glucose tolerance screening
- Gestational Diabetes: Pregnancy-related glucose disorders
- Hypoglycemia: Low blood sugar episodes and patterns
12. USB Digital Stethoscopes
Stethoscope System
| Specification | Details |
|---|---|
| Manufacturer | Thinklabs, StethIO, CardioCommand |
| Device Classification | FDA 510(k) Cleared (selected models) |
| USB Connectivity | Direct audio recording to PC |
| Frequency Range | 20 Hz to 20 kHz |
| Amplification | Up to 100x amplification |
| Recording | Digital audio capture and analysis |
Medical Conditions Detected:
- Heart Murmurs: Systolic and diastolic murmurs
- Lung Sounds: Rales, wheezes, rhonchi
- Cardiac Arrhythmias: Irregular heart rhythms
- Vascular Bruits: Arterial stenosis detection
- Abdominal Sounds: Bowel sounds, vascular bruits
13. USB Medical Scales
Precision Scale
| Specification | Details |
|---|---|
| Manufacturer | Detecto, Health o meter, Tanita |
| Device Classification | FDA 510(k) Cleared |
| USB Features | Weight data transfer to EMR systems |
| Capacity | 400-1000 lbs (180-450 kg) |
| Accuracy | ±0.1 lb (±0.05 kg) |
| Additional Features | BMI calculation, body composition |
Medical Conditions Detected:
- Obesity: BMI calculation and weight management
- Malnutrition: Underweight conditions and monitoring
- Fluid Status: Weight changes indicating fluid retention
- Growth Monitoring: Pediatric weight tracking
- Treatment Response: Medication effects on weight
14. USB Peak Flow Meters
Monitoring System
| Specification | Details |
|---|---|
| Manufacturer | Clement Clarke, Ferraris, MicroLife |
| Device Classification | FDA 510(k) Cleared |
| USB Features | Data logging and trend analysis |
| Range | 50-800 L/min |
| Accuracy | ±10 L/min or ±10% |
| Software | Asthma management applications |
Medical Conditions Detected:
- Asthma: Peak flow monitoring and exacerbation detection
- COPD: Chronic obstructive pulmonary disease management
- Bronchospasm: Airway constriction assessment
- Exercise-Induced Asthma: Activity-related breathing difficulties
- Treatment Response: Medication effectiveness evaluation
15. USB Medical Diagnostic Tablets
Diagnostic Workstation
| Specification | Details |
|---|---|
| Manufacturer | Zebra Technologies, Motion Computing, Getac |
| Device Classification | Medical-grade tablets |
| USB Connectivity | Multiple USB 3.0/3.1 ports for medical devices |
| Certifications | IEC 60601-1 medical electrical equipment |
| Display | 10-12″ medical-grade touchscreen |
| Durability | IP65 rated, antimicrobial coating |
Medical Applications:
- Point-of-Care Testing: Bedside diagnostic capabilities
- EMR Integration: Electronic health record access and updates
- Telemedicine: Remote consultation and monitoring
- Medication Management: Digital prescribing and verification
- Patient Education: Interactive health information delivery
SECTION 3: HYBRID AI + USB MEDICAL DEVICES
This section presents 15 advanced medical devices combining both artificial intelligence capabilities and USB connectivity for comprehensive diagnostic solutions with intelligent analysis and seamless data management.
1. AI-Enhanced USB ECG Systems

| Specification | Details |
|---|---|
| Manufacturer | Schiller, EDAN, Bionet |
| Device Classification | FDA 510(k) Cleared |
| AI Features | Automated rhythm analysis, STEMI detection |
| USB Connectivity | Real-time data transmission and storage |
| Leads | 12-lead configuration with AI interpretation |
| Processing Speed | Real-time analysis within 10 seconds |
Medical Conditions Detected:
- Myocardial Infarction: AI-powered STEMI and NSTEMI detection
- Arrhythmias: Atrial fibrillation, ventricular tachycardia, bradycardia
- Conduction Disorders: AV blocks, bundle branch blocks
- Ischemic Changes: ST depression, T-wave abnormalities
- Electrolyte Imbalances: Hyperkalemia, hypokalemia patterns
2. Smart USB Pulse Oximeters with AI

| Specification | Details |
|---|---|
| Manufacturer | Masimo, Wellue, Nonin |
| Device Classification | FDA 510(k) Cleared |
| AI Capabilities | Predictive SpO2 trending, motion artifact reduction |
| USB Features | Charging, data synchronization, software updates |
| Accuracy | ±1% SpO2 with AI enhancement |
| Monitoring | Continuous tracking with intelligent alerts |
Medical Conditions Detected:
- Respiratory Failure: Hypoxemia with predictive analytics
- Sleep Disorders: Sleep apnea, oxygen desaturation events
- COPD Exacerbations: Early detection of disease progression
- Post-Operative Monitoring: Surgical recovery assessment
- Cardiac Conditions: Heart failure with reduced oxygen delivery
3. AI-Powered USB Spirometers

| Specification | Details |
|---|---|
| Manufacturer | AioCare, Sparo Labs, MIR |
| Device Classification | FDA 510(k) Cleared, CE Marked |
| AI Technology | Automated quality control, cough detection, pattern recognition |
| USB Connectivity | Data transfer, software updates, charging |
| Tests | FVC, FEV1, PEF with AI-enhanced accuracy |
| Platform | Cross-platform compatibility |
Medical Conditions Detected:
- Asthma: AI-powered exacerbation prediction and monitoring
- COPD: Disease progression tracking with machine learning
- Pulmonary Fibrosis: Restrictive pattern recognition
- Neuromuscular Diseases: Respiratory muscle weakness assessment
- Occupational Lung Disease: Work-related exposure monitoring
4. USB-Connected AI Ultrasound Probes

| Specification | Details |
|---|---|
| Manufacturer | Clarius, Butterfly, Philips |
| Device Classification | FDA 510(k) Cleared |
| AI Features | Auto-measurements, needle guidance, pathology detection |
| USB Connectivity | Direct connection to workstations, data transfer |
| Probe Types | Linear, curved, phased array with AI enhancement |
| Image Processing | Real-time AI image optimization |
Medical Conditions Detected:
- Cardiac Abnormalities: AI-assisted ejection fraction calculation
- Vascular Disease: Automated stenosis detection
- Abdominal Pathology: Organ size measurement, fluid detection
- MSK Injuries: Tendon tears, joint effusions with AI analysis
- Emergency Conditions: FAST exam with intelligent interpretation
5. AI Blood Glucose Monitors with USB

| Specification | Details |
|---|---|
| Manufacturer | iHealth, Dexcom, Abbott |
| Device Classification | FDA 510(k) Cleared |
| AI Capabilities | Predictive glucose trending, pattern recognition |
| USB Features | Data synchronization, software updates, charging |
| Integration | CGM data fusion with AI algorithms |
| Alerts | Intelligent hypoglycemia/hyperglycemia prediction |
Medical Conditions Detected:
- Type 1 Diabetes: AI-powered insulin dosing recommendations
- Type 2 Diabetes: Lifestyle intervention optimization
- Pre-diabetes: Early detection and progression monitoring
- Gestational Diabetes: Pregnancy-specific glucose management
- Hypoglycemic Episodes: Predictive low glucose alerting
6. USB AI Stethoscopes

| Specification | Details |
|---|---|
| Manufacturer | Eko Health, 3M Littmann, Thinklabs |
| Device Classification | FDA 510(k) Cleared |
| AI Technology | Heart murmur detection, lung sound analysis |
| USB Features | Audio recording, data transfer, firmware updates |
| Amplification | 40x digital amplification with AI enhancement |
| Recording | High-fidelity digital audio capture |
Medical Conditions Detected:
- Heart Murmurs: AI-powered pathological murmur detection
- Atrial Fibrillation: Irregular rhythm identification
- Heart Failure: S3/S4 gallop detection
- Valvular Disease: Stenosis and regurgitation sounds
- Pulmonary Conditions: Rales, wheezes, stridor detection
7. AI-Enhanced USB Thermometers

| Specification | Details |
|---|---|
| Manufacturer | Withings, ThermoWorks, Braun |
| Device Classification | FDA 510(k) Cleared (selected models) |
| AI Features | Smart fever pattern recognition, thermal imaging AI |
| USB Connectivity | Data logging, charging, software updates |
| Accuracy | ±0.1°C with AI compensation |
| Measurement Time | 1-3 seconds with intelligent positioning |
Medical Conditions Detected:
- Infectious Diseases: Fever pattern analysis for early detection
- COVID-19 Screening: Temperature-based triage
- Sepsis: Fever/hypothermia patterns with AI analysis
- Inflammatory Conditions: Temperature trend monitoring
- Drug Reactions: Medication-induced hyperthermia
8. USB AI Blood Pressure Monitors

| Specification | Details |
|---|---|
| Manufacturer | OMRON, A&D Medical, Welch Allyn |
| Device Classification | FDA 510(k) Cleared |
| AI Technology | Hypertension prediction, irregular heartbeat detection |
| USB Features | Data synchronization, charging, EMR integration |
| Measurements | BP, ECG, AFib detection with AI analysis |
| Memory | Unlimited cloud storage with AI trending |
Medical Conditions Detected:
- Hypertension: AI-powered blood pressure trend analysis
- White Coat Syndrome: Environmental BP variation detection
- Masked Hypertension: Hidden high blood pressure identification
- Atrial Fibrillation: Irregular pulse detection during BP measurement
- Cardiovascular Risk: AI-based risk stratification
9. AI-Powered USB Medical Imaging
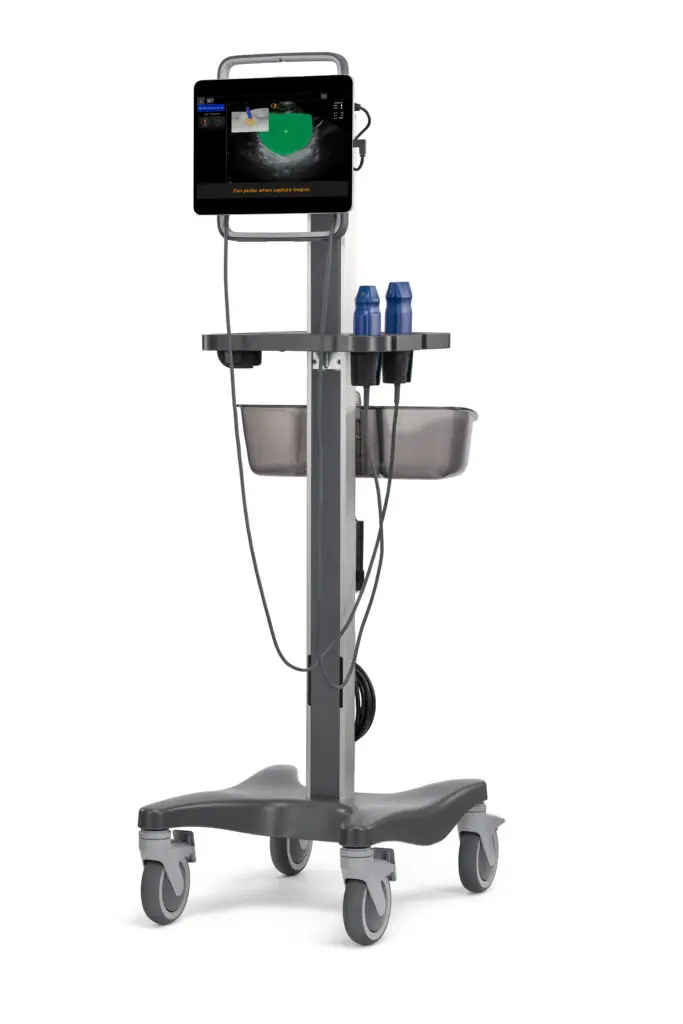
| Specification | Details |
|---|---|
| Manufacturer | EchoNous, Exo, Butterfly Network |
| Device Classification | FDA 510(k) Cleared |
| AI Features | Automated pathology detection, image enhancement |
| USB Connectivity | High-speed data transfer, workstation integration |
| Imaging Modes | B-mode, Doppler, M-mode with AI optimization |
| Processing | Edge AI computing for real-time analysis |
Medical Conditions Detected:
- Cardiac Pathology: AI-assisted valve disease, wall motion abnormalities
- Vascular Disease: Automated stenosis quantification
- Emergency Medicine: FAST protocol with AI interpretation
- Obstetric Conditions: Fetal abnormality detection
- Abdominal Emergencies: Free fluid, organ pathology detection
10. Smart USB Health Monitoring Hubs

| Specification | Details |
|---|---|
| Manufacturer | Withings, Philips Healthcare, GE HealthCare |
| Device Classification | FDA 510(k) Cleared (components) |
| AI Integration | Multi-parameter AI analysis and correlation |
| USB Features | Device charging hub, data aggregation |
| Parameters | Vital signs, biometrics, environmental data |
| Connectivity | Wi-Fi, Bluetooth, USB 3.0, Ethernet |
Medical Conditions Detected:
- Chronic Disease Management: Multi-parameter trend analysis
- Early Warning Systems: AI-powered deterioration prediction
- Medication Adherence: Treatment compliance monitoring
- Risk Stratification: Multi-modal health risk assessment
- Population Health: Aggregate health pattern analysis
11. AI-Enhanced USB Respiratory Monitors

| Specification | Details |
|---|---|
| Manufacturer | Sparo Labs, AioCare, MIR |
| Device Classification | FDA 510(k) Cleared |
| AI Technology | Breathing pattern analysis, predictive exacerbation |
| USB Connectivity | Continuous data streaming, charging |
| Monitoring | Real-time respiratory rate, effort, quality |
| Alerts | AI-powered early warning system |
Medical Conditions Detected:
- Respiratory Failure: AI-predicted respiratory compromise
- Sleep Apnea: Breathing pattern abnormality detection
- COPD Monitoring: Disease progression tracking
- Asthma Management: Exacerbation prediction and prevention
- Post-Operative Care: Respiratory recovery monitoring
12. USB AI Cardiac Rhythm Monitors
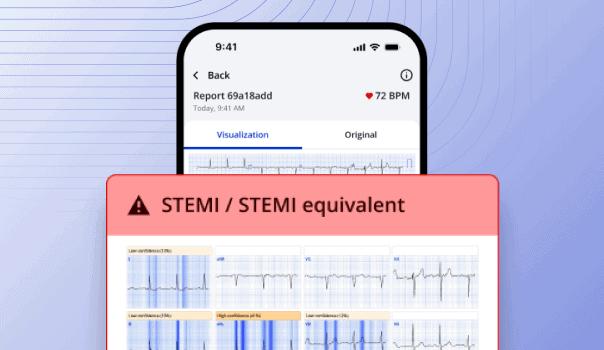
| Specification | Details |
|---|---|
| Manufacturer | Powerful Medical, iRhythm, BioTelemetry |
| Device Classification | FDA 510(k) Cleared |
| AI Algorithms | Advanced arrhythmia detection and classification |
| USB Features | High-speed ECG data upload, analysis results |
| Monitoring Duration | Continuous 24/7 rhythm analysis |
| Detection Rate | 99.7% accuracy for critical arrhythmias |
Medical Conditions Detected:
- Atrial Fibrillation: AI-powered AFib burden calculation
- Ventricular Arrhythmias: VT/VF detection and analysis
- Bradyarrhythmias: Sinus node dysfunction, heart blocks
- Syncope Evaluation: Rhythm-related fainting episodes
- Pacemaker Assessment: Device malfunction detection
13. AI-Powered USB Diagnostic Workstations

| Specification | Details |
|---|---|
| Manufacturer | GE HealthCare, Philips, Siemens Healthineers |
| Device Classification | FDA 510(k) Cleared |
| AI Integration | Multi-modal diagnostic AI platform |
| USB Connectivity | Multiple device integration, data fusion |
| Processing Power | GPU-accelerated AI inference engine |
| Interoperability | HL7 FHIR, DICOM, EMR integration |
Medical Applications:
- Comprehensive Diagnostics: Multi-parameter AI analysis
- Clinical Decision Support: Evidence-based recommendations
- Risk Assessment: Predictive analytics for patient outcomes
- Workflow Optimization: AI-powered clinical efficiency
- Quality Assurance: Automated diagnostic verification
14. Smart USB Medical Data Loggers

| Specification | Details |
|---|---|
| Manufacturer | Wellue, Nonin, Masimo |
| Device Classification | FDA 510(k) Cleared |
| AI Features | Pattern recognition, anomaly detection |
| USB Connectivity | High-capacity data storage and transfer |
| Memory | Up to 1TB storage with compression |
| Battery Life | 30+ days continuous monitoring |
Medical Applications:
- Ambulatory Monitoring: Extended patient observation
- Clinical Research: Long-term data collection
- Remote Monitoring: Home-based patient care
- Trend Analysis: AI-powered health pattern recognition
- Medication Studies: Drug efficacy and safety monitoring
15. AI-Integrated USB Telemedicine Kits
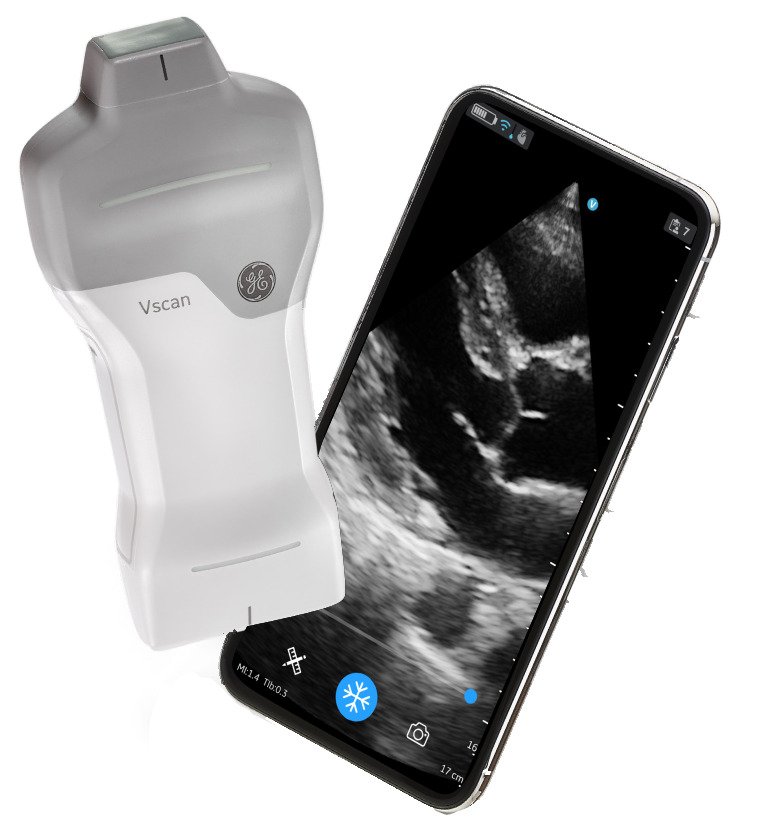
| Specification | Details |
|---|---|
| Manufacturer | AMD Global Telemedicine, Tyto Care, GlobalMed |
| Device Classification | FDA 510(k) Cleared (components) |
| AI Capabilities | Automated examination guidance, diagnostic assistance |
| USB Hub | Central connectivity for all diagnostic devices |
| Components | Stethoscope, otoscope, camera, thermometer, scales |
| Connectivity | 4K video streaming, real-time data transmission |
Medical Applications:
- Remote Consultations: AI-guided physical examinations
- Rural Healthcare: Specialist access via telemedicine
- Chronic Care Management: Regular monitoring and follow-up
- Emergency Triage: AI-powered severity assessment
- Health Screening: Preventive care and early detection
CONCLUSION
Summary of 45 Medical Devices: This comprehensive documentation has presented 45 cutting-edge medical devices across three categories: 15 AI-enabled devices, 15 USB-connected devices, and 15 hybrid AI+USB systems. These devices represent the forefront of modern healthcare technology, offering unprecedented diagnostic capabilities, improved patient outcomes, and enhanced clinical workflows.
Technology Integration: The convergence of artificial intelligence and Universal Serial Bus connectivity is revolutionizing healthcare delivery. AI provides predictive analytics, automated diagnosis, and clinical decision support, while USB connectivity ensures seamless data management, real-time monitoring, and interoperability with healthcare information systems.
Clinical Impact: These devices collectively address a comprehensive range of medical conditions including cardiovascular disease, respiratory disorders, diabetes, infectious diseases, neurological conditions, and emergency medicine scenarios. The integration of AI and USB technologies enables earlier detection, more accurate diagnosis, and personalized treatment approaches.
Future Outlook: The medical device industry continues to evolve rapidly with advancing AI capabilities, improved sensor technologies, and enhanced connectivity options. Healthcare professionals can expect continued innovation in point-of-care diagnostics, remote monitoring solutions, and intelligent clinical decision support systems.
Regulatory Compliance: All devices presented in this documentation maintain appropriate regulatory clearances including FDA 510(k) approvals, CE marking, and other international certifications, ensuring safety and efficacy for clinical use.