COMPLETE USB MEDICAL EQUIPMENT CATALOG
Comprehensive Guide to 50+ USB-Connected Medical Devices from Leading Global Manufacturers
MEDICAL DEVICE CATEGORIES
Pulse Oximeters & SpO2 Monitoring
Nonin Model 3231 USB Pulse Oximeter
Company: Nonin Medical, Inc.
Location: 13700 1st Avenue North, Plymouth, Minnesota 55441, USA
Founded: 1986
Detects & Monitors:
Technical Specifications:
Masimo Rad-67 Pulse Co-Oximeter
Company: Masimo Corporation
Location: Irvine, California, USA
Founded: 1989
Detects & Monitors:
Technical Specifications:
CONTEC CMS50D USB
CONTEC CMS50D USB Pulse Oximeter
Company: Contec Medical Systems Co., Ltd.
Location: Qinhuangdao, Hebei Province, China
Founded: 1996
Detects & Monitors:
Technical Specifications:
ECG/EKG & Cardiac Monitoring Devices
3M™ Littmann® CORE Digital Stethoscope
Partnership: 3M Company (USA) + Eko Health (California)
Technology: Advanced digital auscultation
Detects & Monitors:
Technical Specifications:
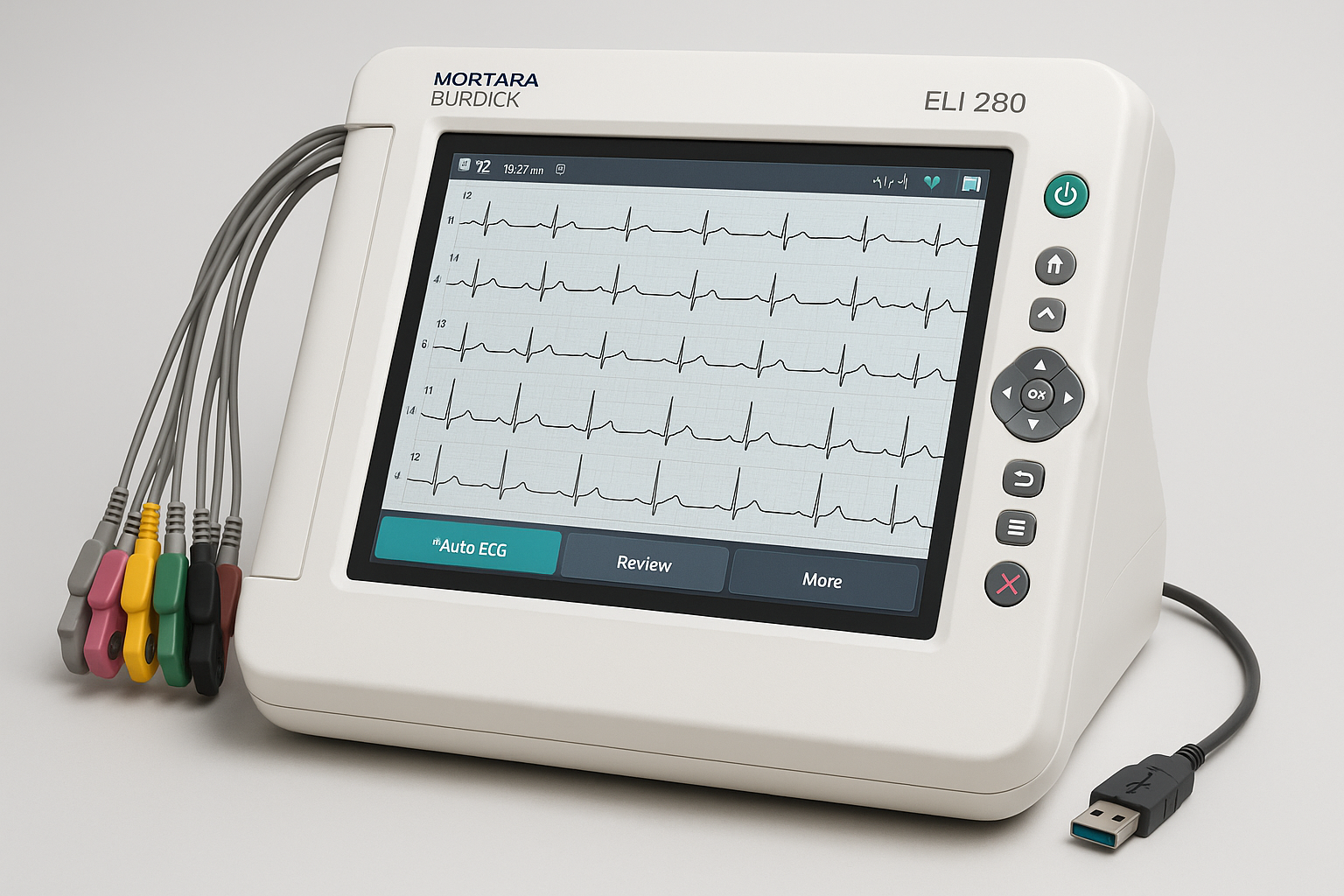
Mortara Burdick ELI 280 ECG System
Company: Mortara Instrument, Inc.
Location: Milwaukee, Wisconsin, USA
Specialty: Cardiac diagnostic equipment
Detects & Monitors:
Technical Specifications:
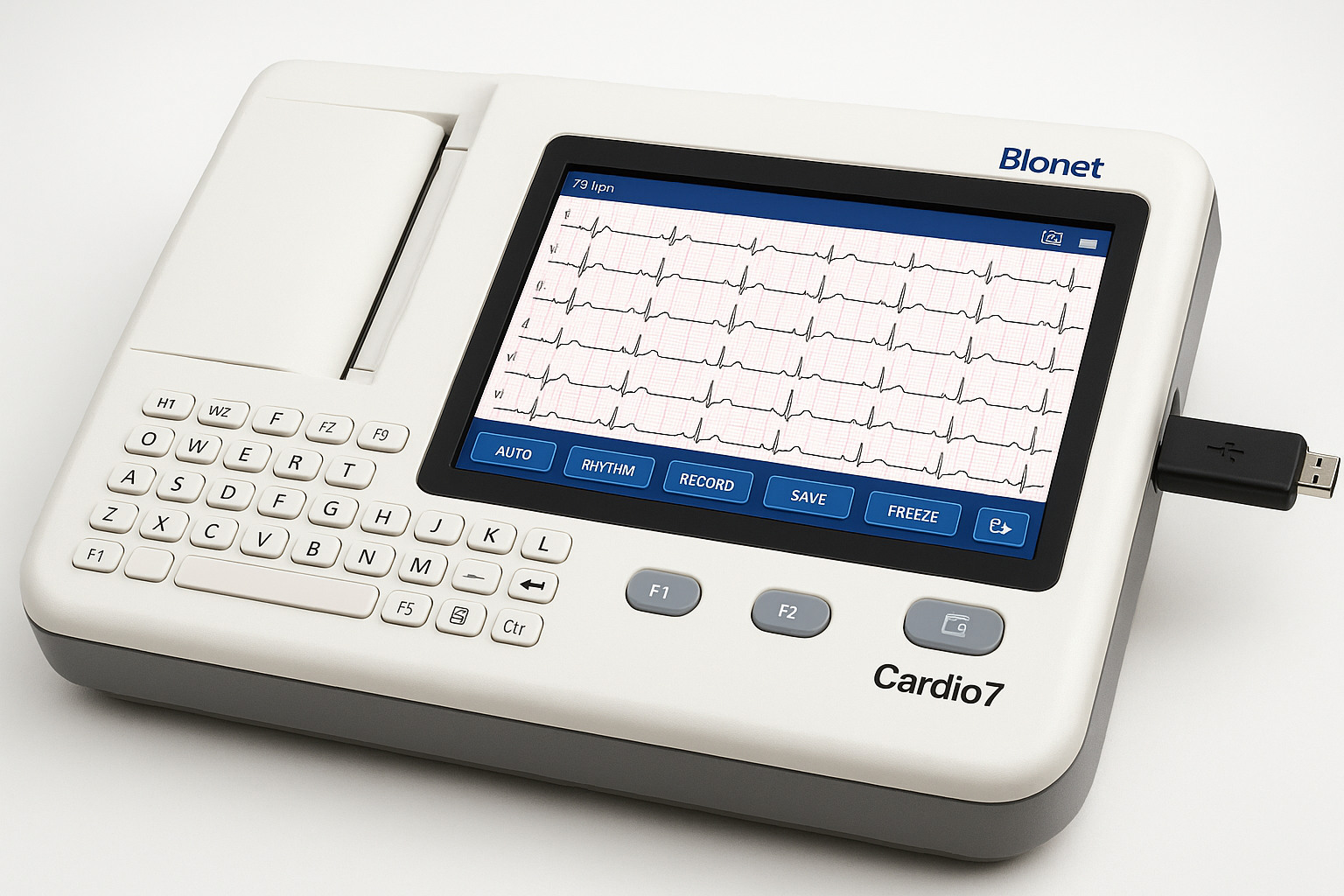
Bionet Cardio7 Touch Screen ECG
Company: Bionet America, Inc.
Location: Tustin, California, USA
Parent: Bionet Co., Ltd. (South Korea)
Detects & Monitors:
Technical Specifications:
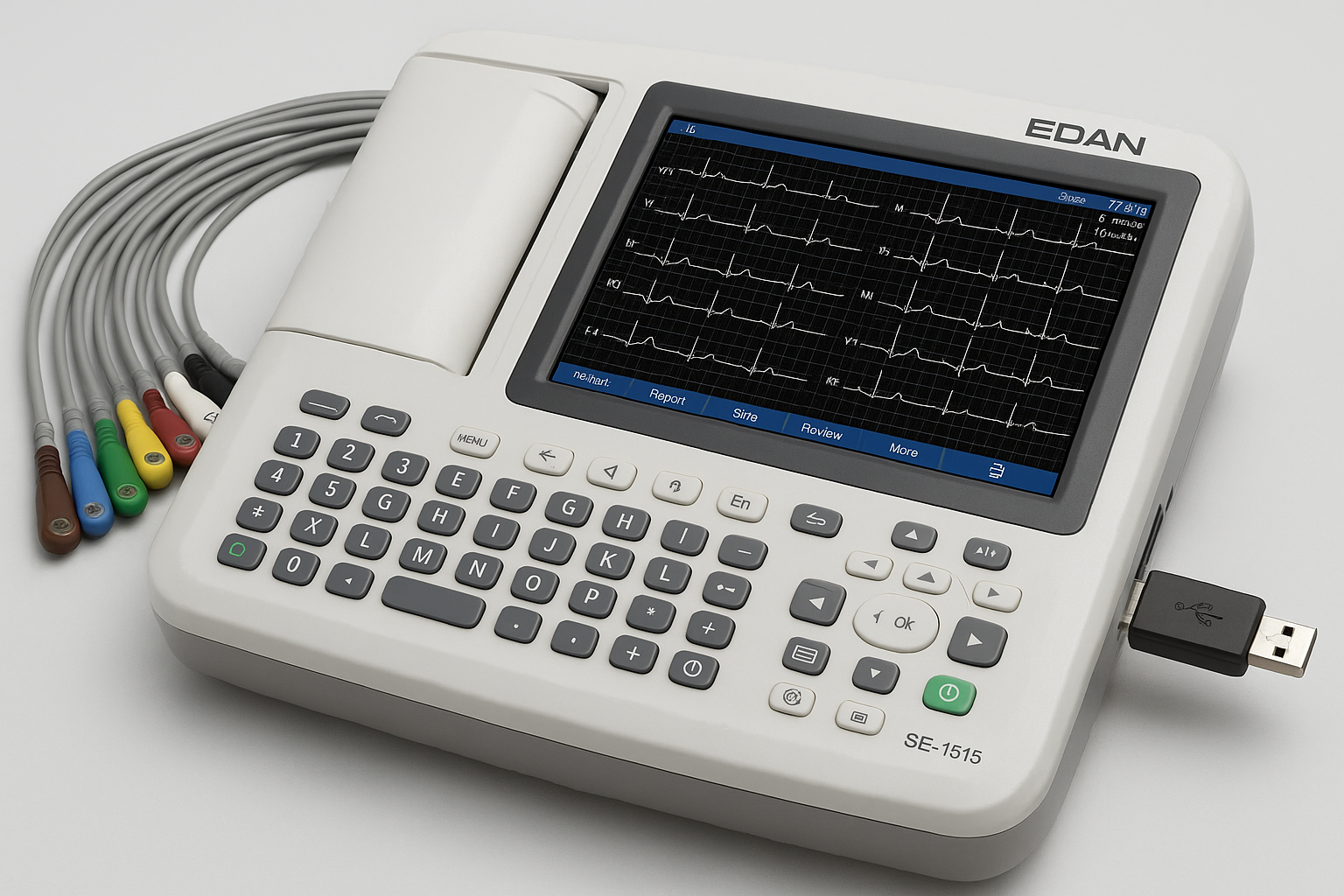
Edan SE-1515 15-Lead ECG System
Company: Edan Instruments, Inc.
Location: Shenzhen, China
Founded: 1995
Detects & Monitors:
Technical Specifications:
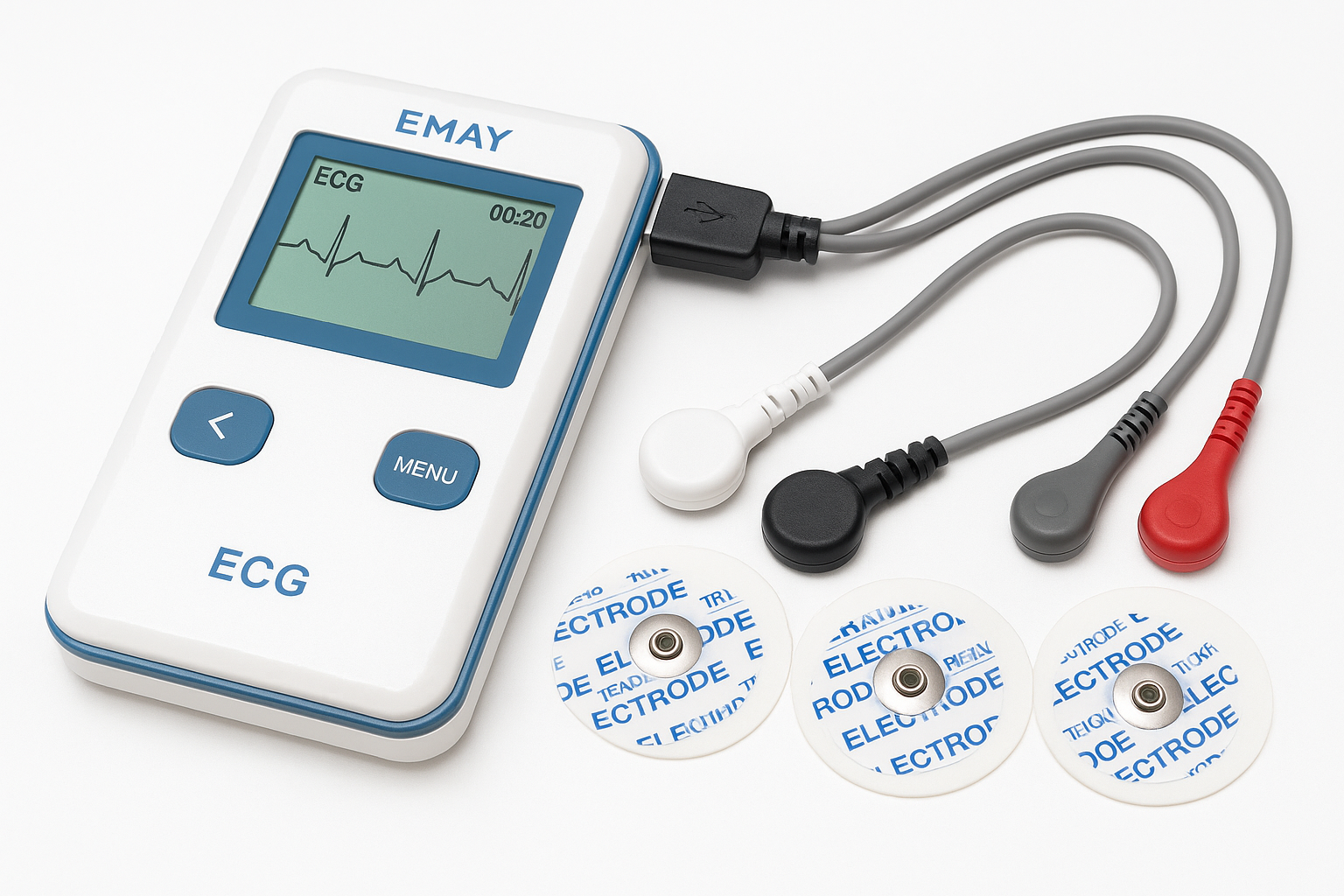
EMAY Portable ECG Monitor
Company: EMAY Limited
Location: Hong Kong
Specialty: Portable cardiac monitoring
Detects & Monitors:
Technical Specifications:
Blood Pressure & Vital Signs Monitoring
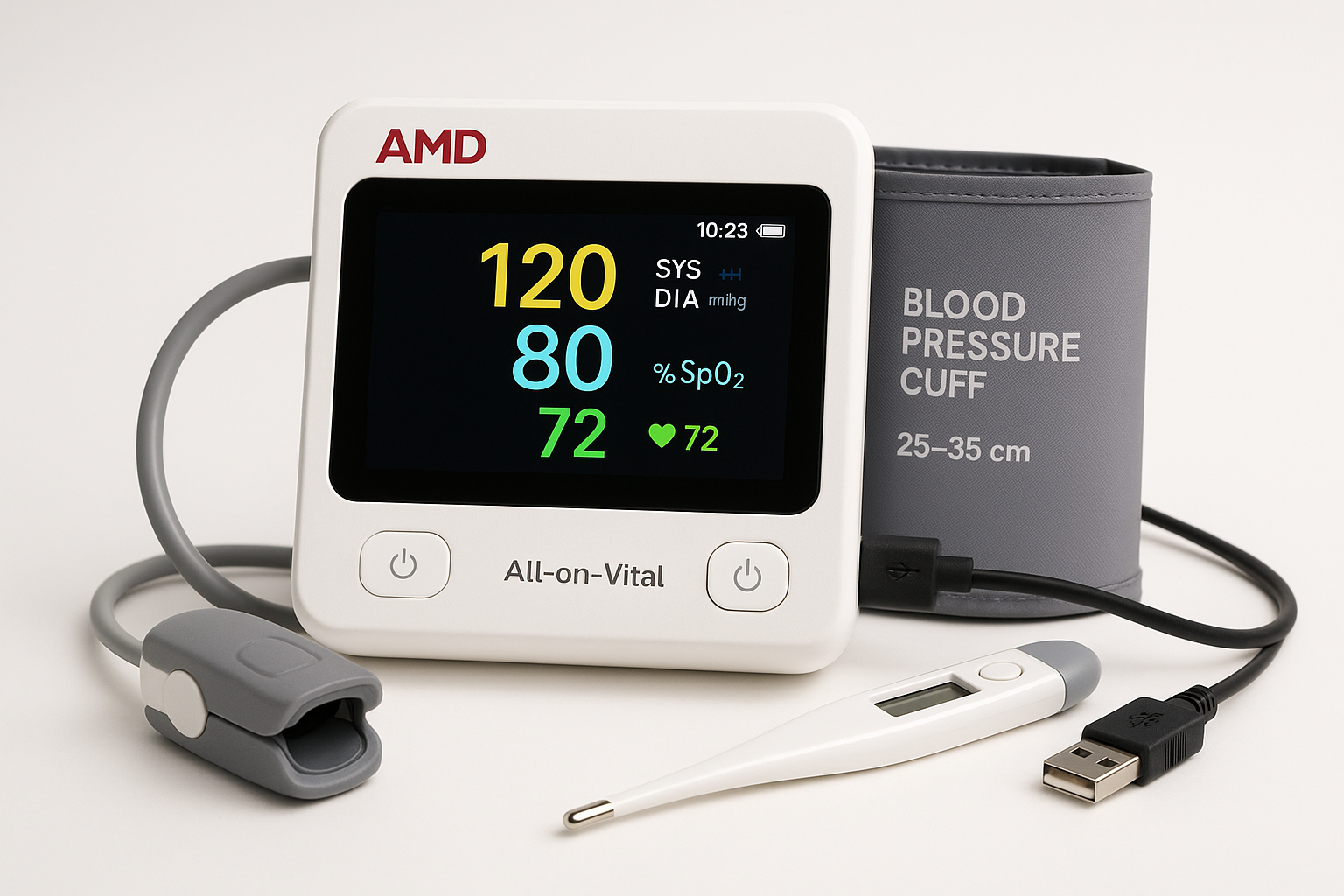
AMD All-in-One Vital Signs Monitor
Company: AMD Global Telemedicine, Inc.
Location: Chelmsford, Massachusetts, USA
Founded: 1991
Detects & Monitors:
Technical Specifications:
Omron HeartGuide Wearable BP Monitor
Company: Omron Healthcare, Inc.
Location: Lake Forest, Illinois, USA
Parent: Omron Corporation (Japan)
Detects & Monitors:
Technical Specifications:
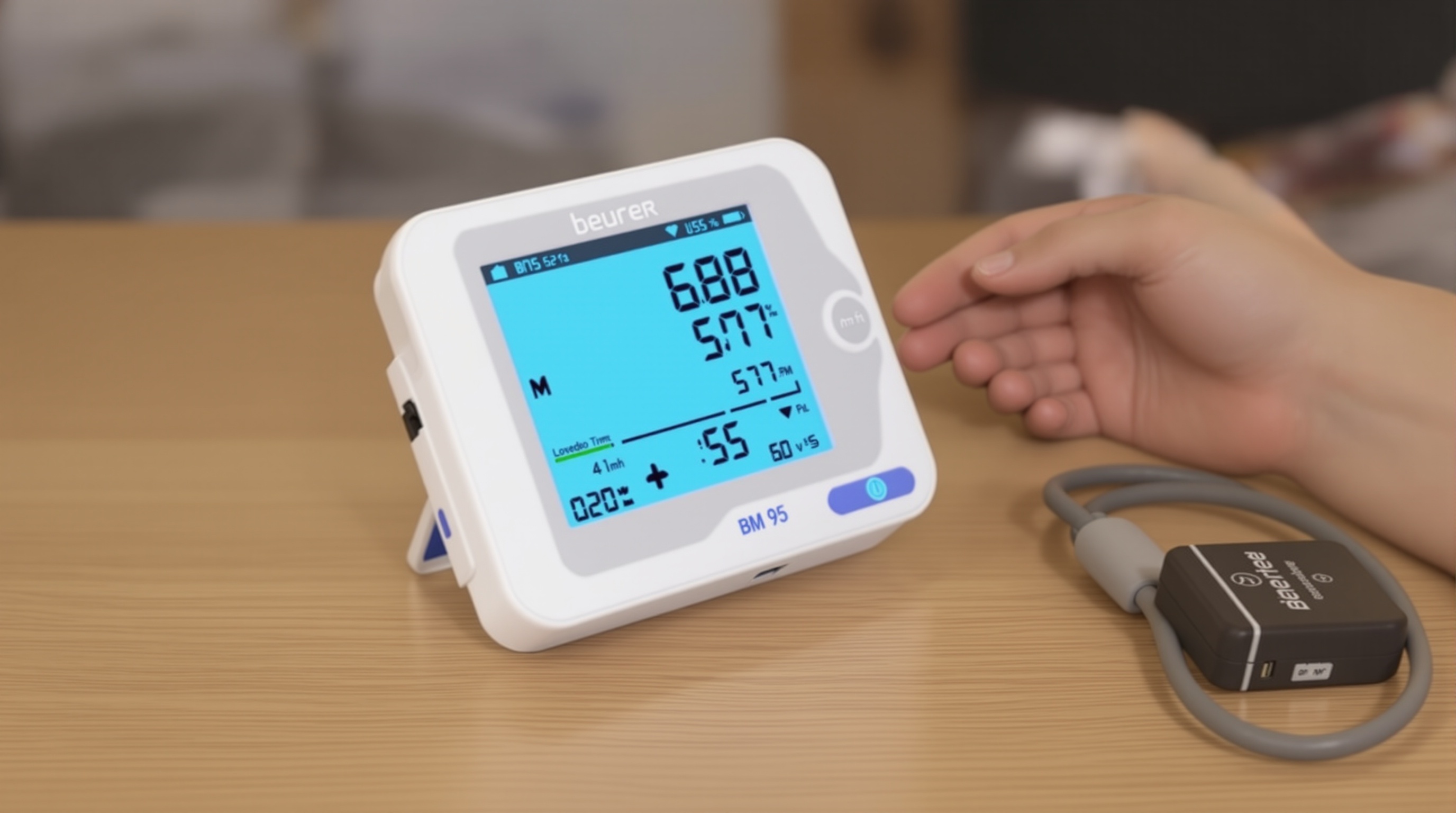
Beurer BM 95 Upper Arm BP Monitor
Company: Beurer GmbH
Location: Ulm, Germany
Founded: 1919
Detects & Monitors:
Technical Specifications:
Spirometry & Pulmonary Function Testing
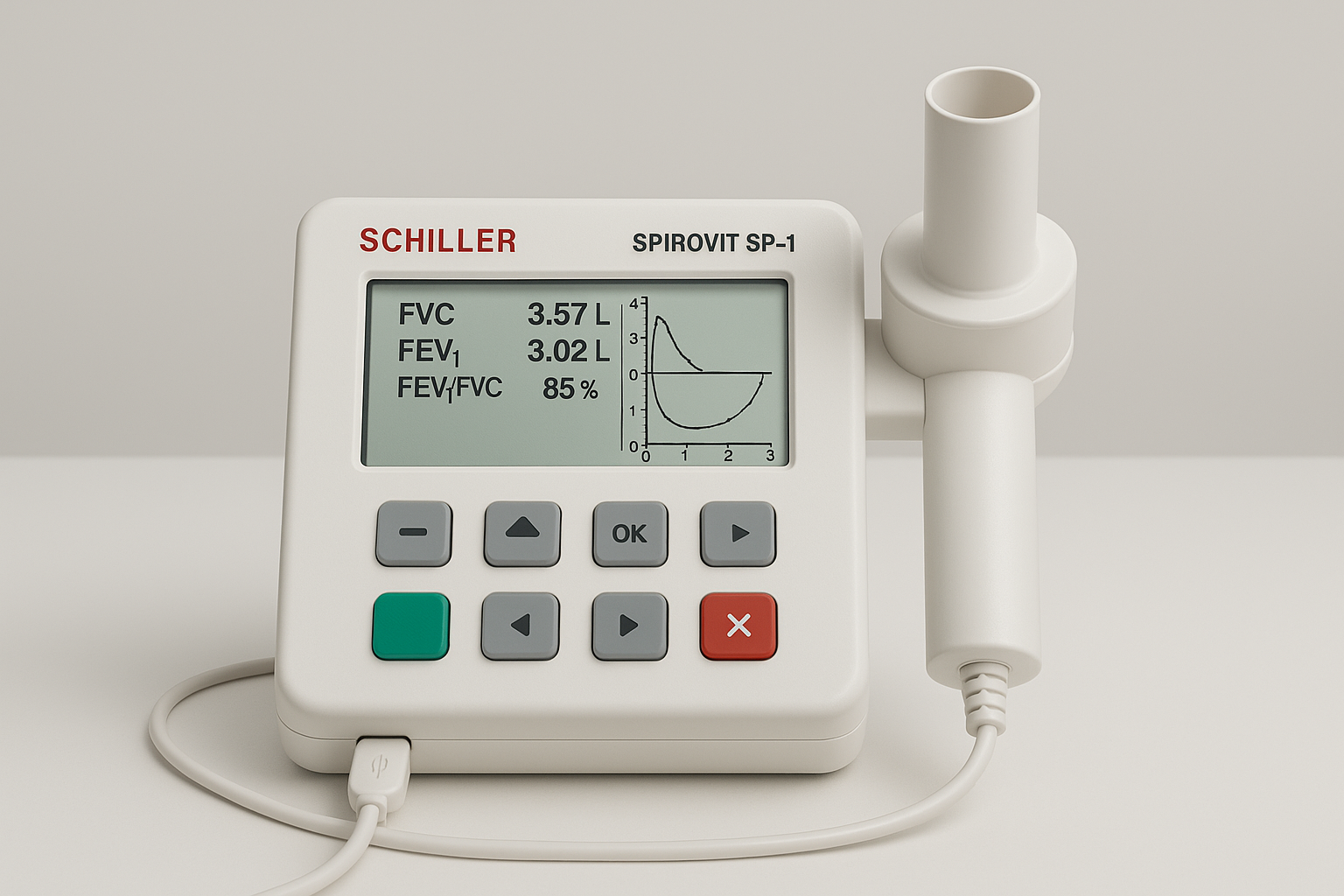
SCHILLER SPIROVIT SP-1 Spirometer
Company: SCHILLER AG
Location: Baar, Switzerland
Founded: 1974
Detects & Monitors:
Technical Specifications:
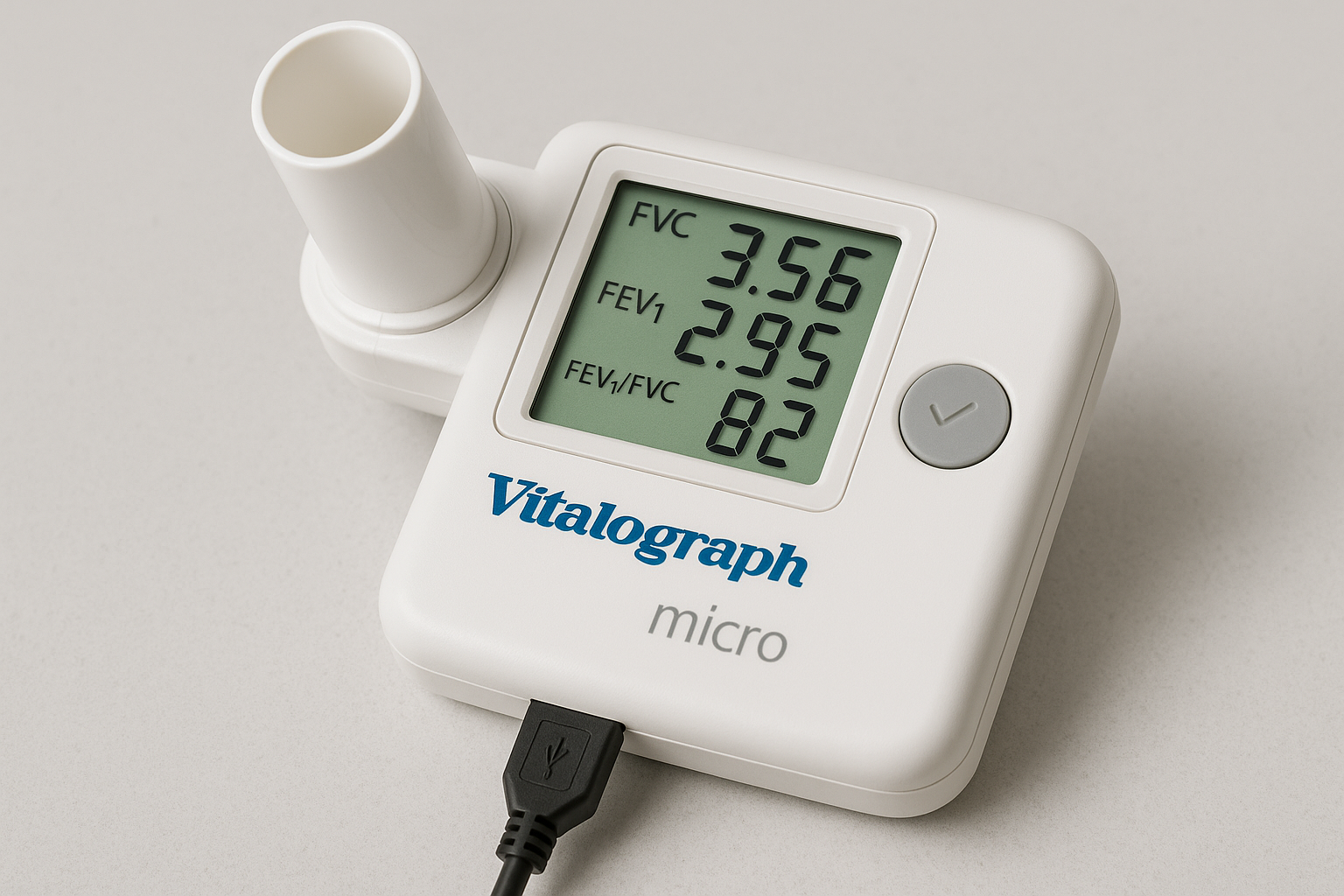
Vitalograph Micro Spirometer
Company: Vitalograph Ltd.
Location: Buckingham, England, UK
Founded: 1963
Detects & Monitors:
Technical Specifications:
NDD EasyOne Air Spirometer
Company: ndd Medizintechnik AG
Location: Zurich, Switzerland
Founded: 1988
Detects & Monitors:
Technical Specifications:
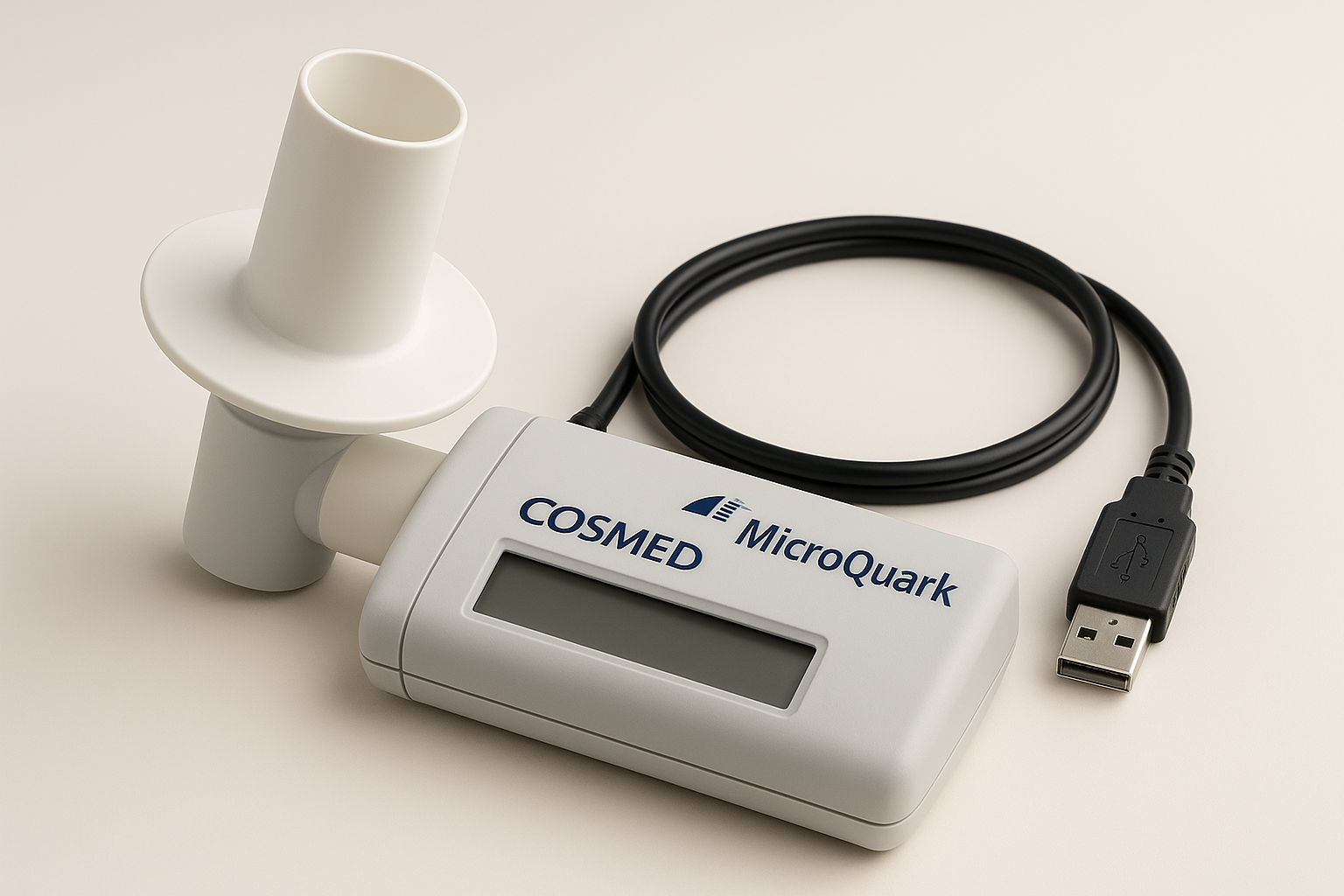
COSMED MicroQuark Spirometer
Company: COSMED srl
Location: Rome, Italy
Founded: 1980
Detects & Monitors:
Technical Specifications:
Digital Thermometers & Temperature Monitoring
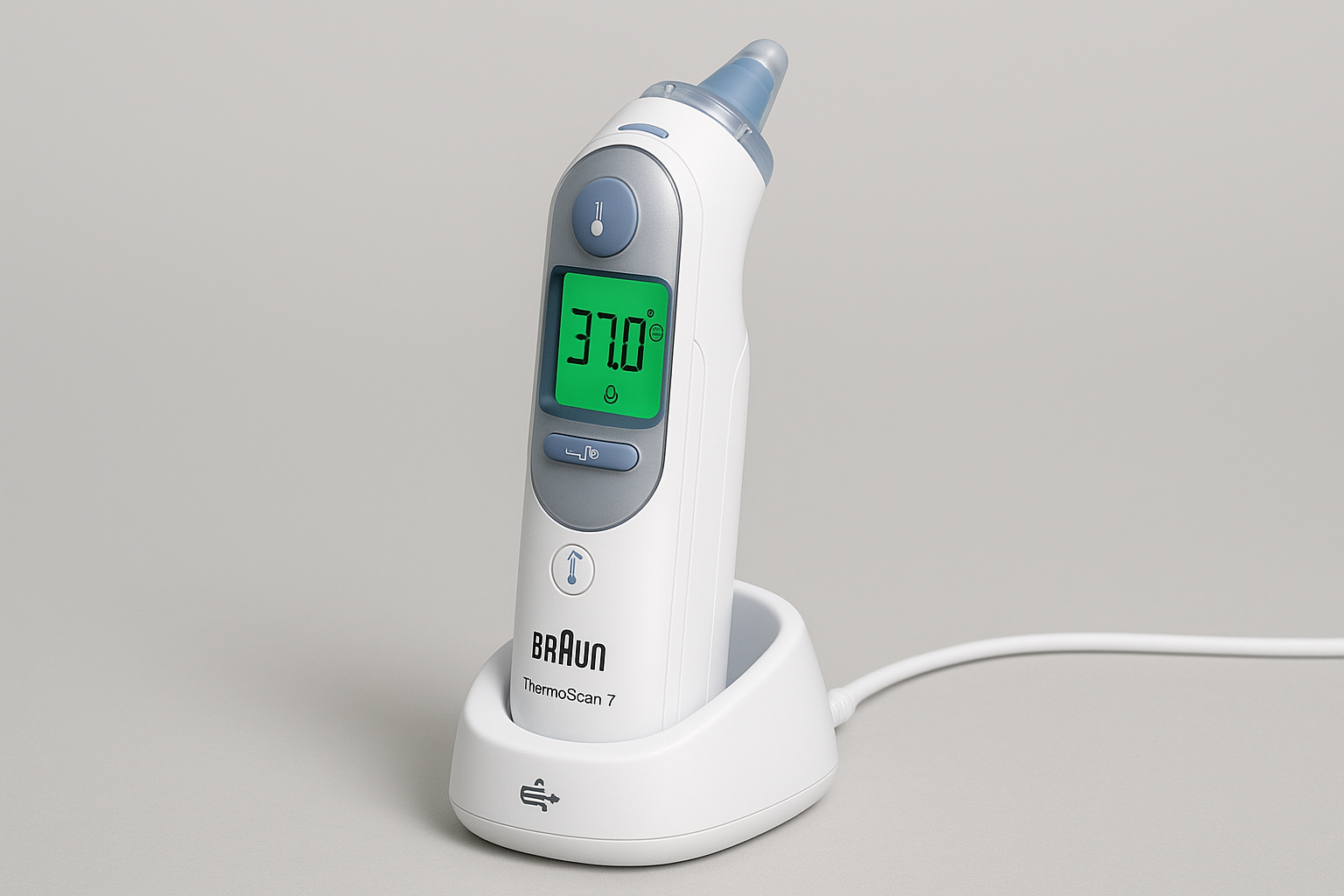
Braun ThermoScan 7 Ear Thermometer
Company: Braun Healthcare (Procter & Gamble)
Location: Kronberg, Germany
Founded: 1921
Detects & Monitors:
Technical Specifications:
Exergen TAT-5000 Temporal Artery Thermometer
Company: Exergen Corporation
Location: Watertown, Massachusetts, USA
Founded: 1980
Detects & Monitors:
Technical Specifications:
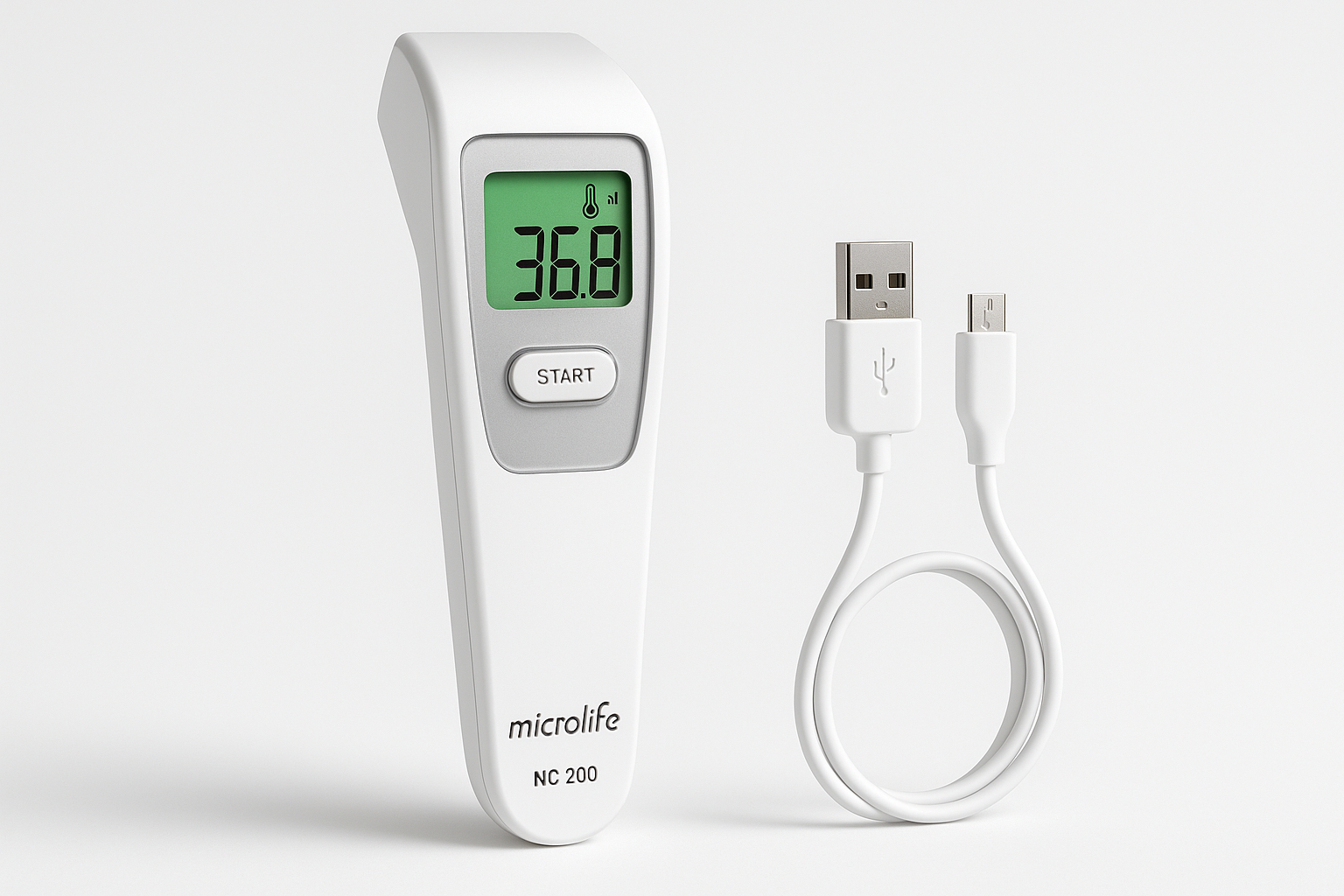
Microlife NC 200 Digital Thermometer
Company: Microlife Corporation
Location: Widnau, Switzerland
Founded: 1981
Detects & Monitors:
Technical Specifications:
Glucose Meters & Diabetes Management
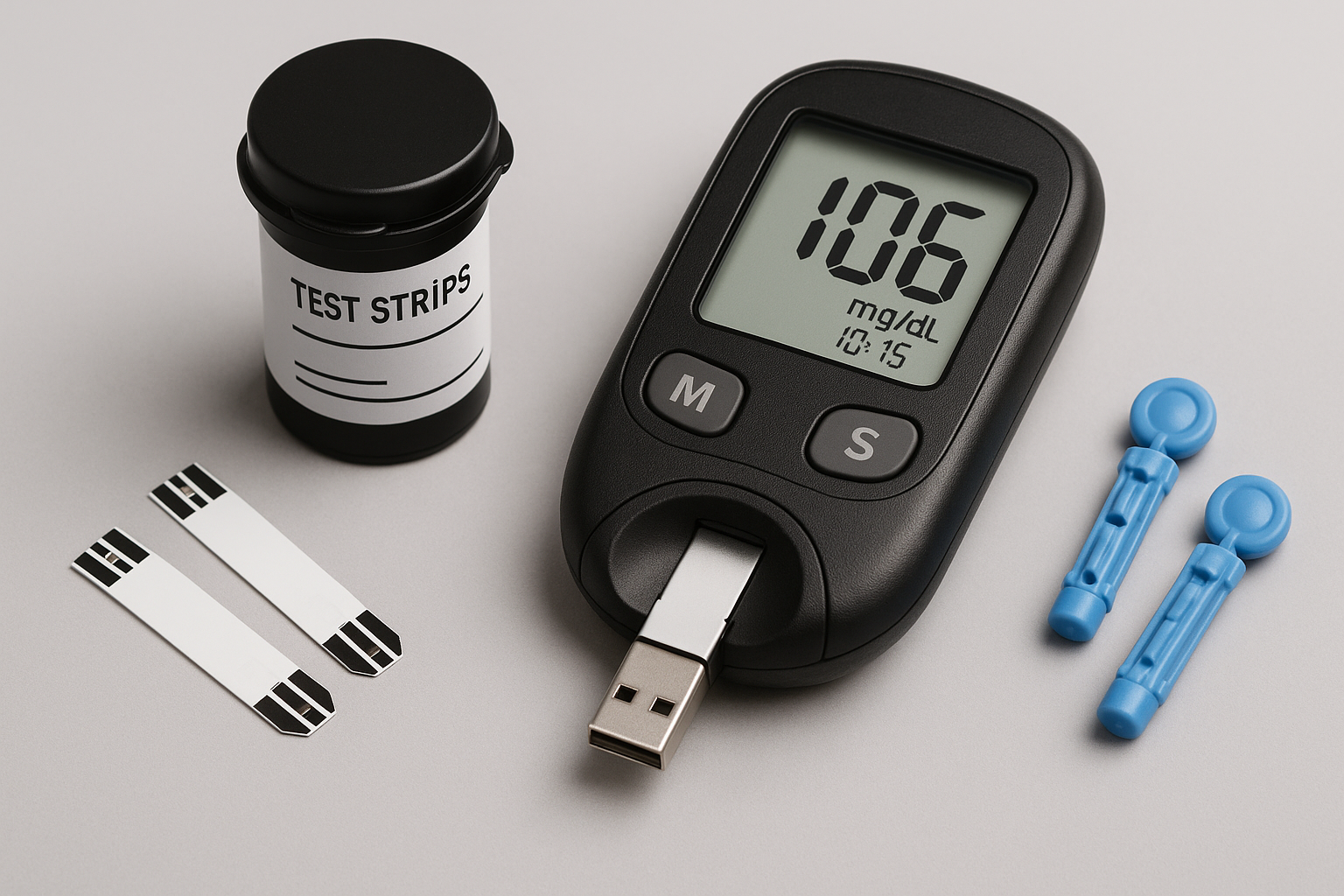
Accu-Chek® Guide Glucose Meter
Company: Roche Diabetes Care, Inc.
Parent: Roche Holding AG, Switzerland
Location: Indianapolis, Indiana, USA
Detects & Monitors:
Technical Specifications:
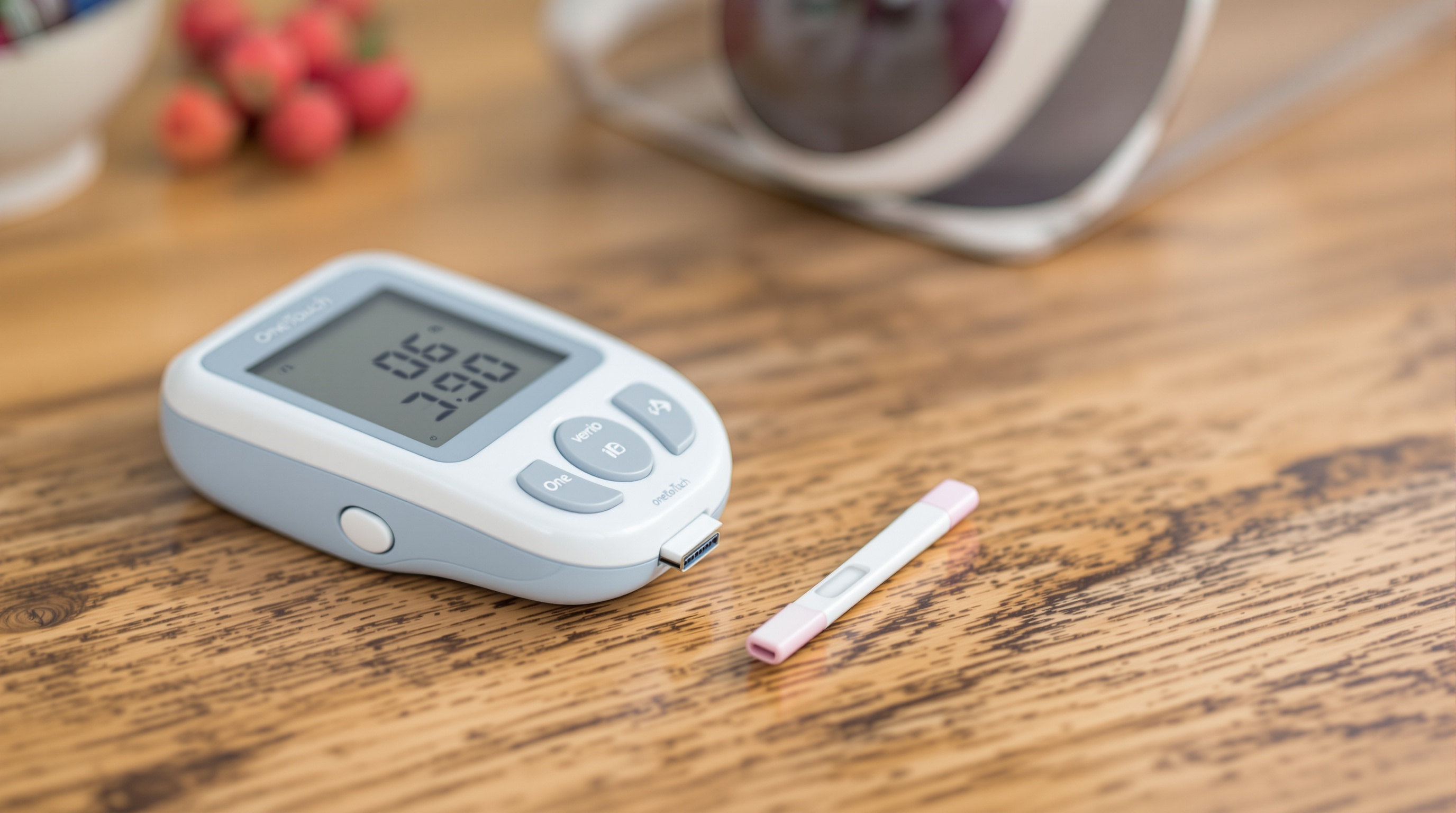
OneTouch Verio Glucose Meter
Company: LifeScan, Inc. (Johnson & Johnson)
Location: Malvern, Pennsylvania, USA
Founded: 1986
Detects & Monitors:
Technical Specifications:
FreeStyle Libre Continuous Glucose Monitor
Company: Abbott Laboratories
Location: Abbott Park, Illinois, USA
Founded: 1888
Detects & Monitors:
Technical Specifications:
Ophthalmology & Eye Examination Devices
HEINE Beta 200 Ophthalmoscope
Company: HEINE Optotechnik GmbH & Co. KG
Location: Herrsching, Germany
Founded: 1946
Detects & Monitors:
Technical Specifications:
Keeler Vantage Plus Ophthalmoscope
Company: Keeler Ltd.
Location: Windsor, Berkshire, UK
Founded: 1917
Detects & Monitors:
Technical Specifications:
Optomed Smartscope PRO Fundus Camera
Company: Optomed Oy
Location: Oulu, Finland
Founded: 2004
Detects & Monitors:
Technical Specifications:
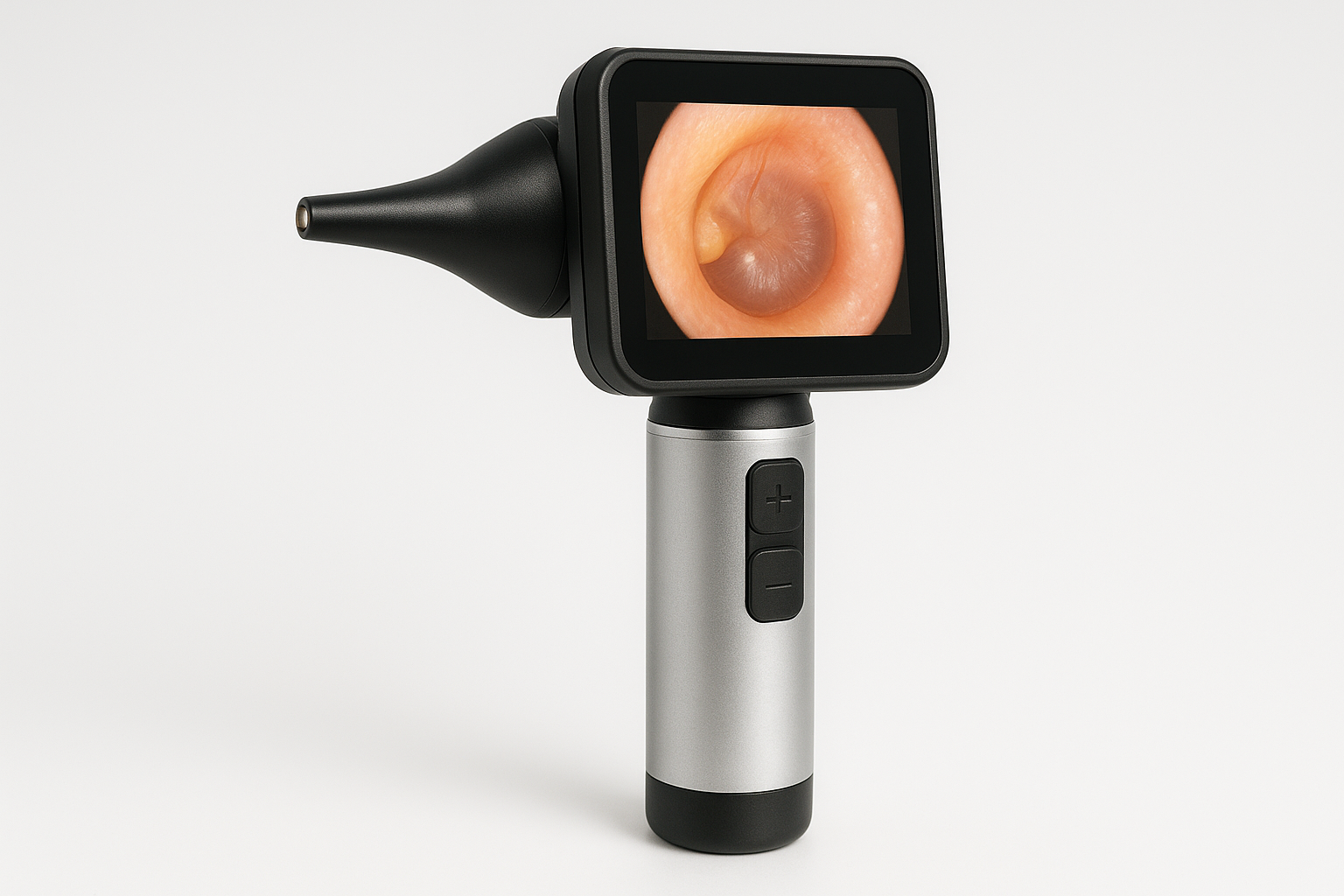
Welch Allyn Digital Otoscope
Company: Welch Allyn (Division of Hill-Rom, now Baxter)
Location: Skaneateles Falls, New York, USA
Founded: 1915
Detects & Monitors:
Technical Specifications:
DermLite DL4/DL5 USB Dermatoscope
Company: 3Gen, LLC
Location: San Juan Capistrano, California, USA
Founded: 1999
Detects & Monitors:
Technical Specifications:
Ultrasound & Imaging Devices
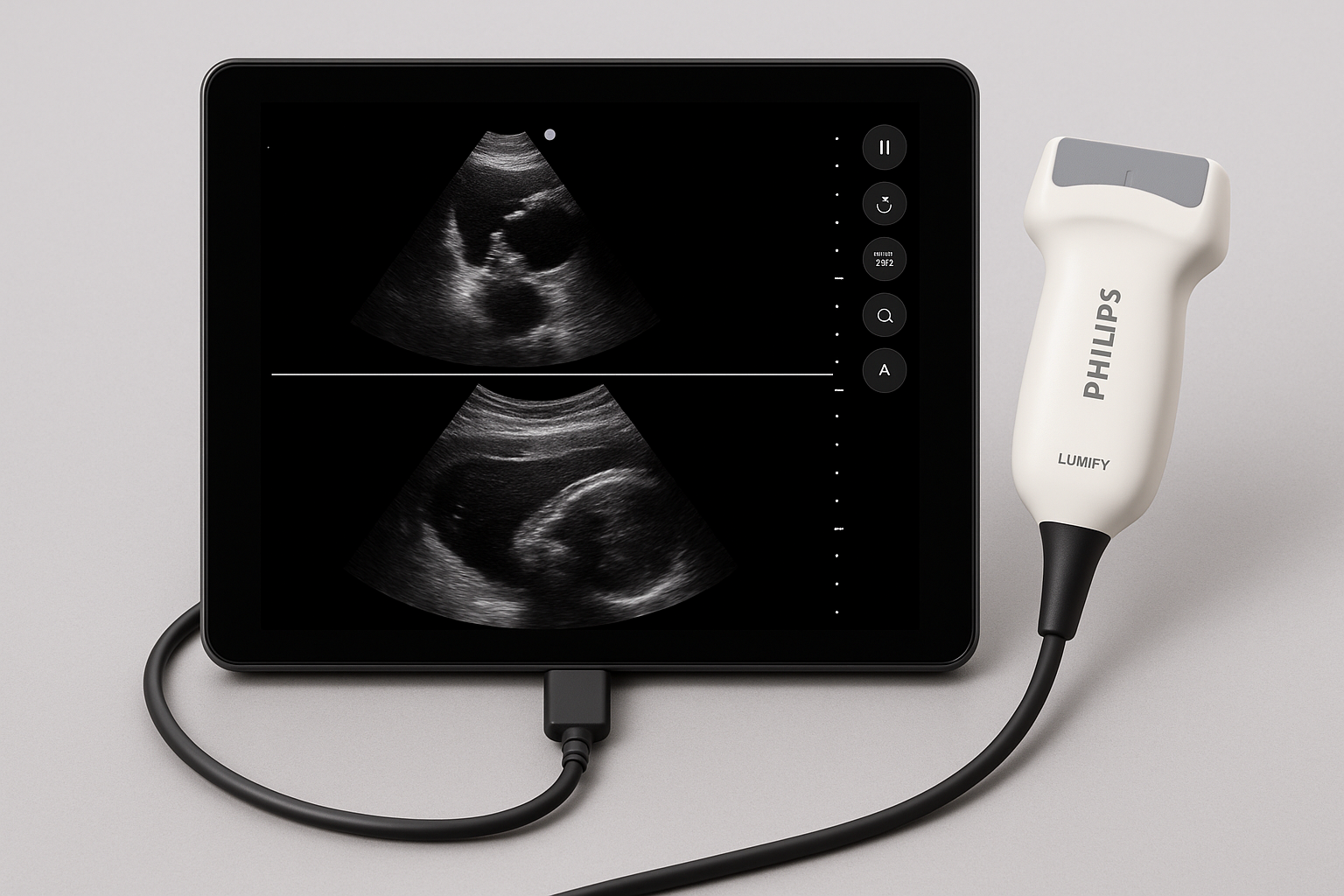
Philips Lumify Handheld Ultrasound
Company: Philips Healthcare
Parent: Royal Philips, Netherlands
Location: Bothell, Washington, USA
Detects & Monitors:
Technical Specifications:
More Devices Available
50+ Additional USB Medical Devices
This catalog includes devices from:
- • GE Healthcare
- • Siemens Healthineers
- • Mindray Medical
- • Boston Scientific
- • Medtronic
- • BD (Becton Dickinson)
- • Vyaire Medical
- • MGC Diagnostics
- + Many more manufacturers