ARCHITECTURAL PARADIGMS IN DIGITAL DENTISTRY:
AI BUILT-IN (EDGE) VS. CLOUD-BASED AI DEVICES
Table of Contents
- 1. Introduction and Executive Summary 1
- 2. Understanding AI Architectures in Dental Technology 3
- 3. AI Built-in Dental Devices: Deep Dive 5
- 4. Cloud-Based AI Dental Devices: Deep Dive 8
- 5. Comparative Analysis 11
- 6. Future Trends and Hybrid Solutions 14
- 7. Conclusion and Recommendations 16
1. Introduction and Executive Summary
The integration of Artificial Intelligence (AI) into dentistry has transcended the phase of novelty to become a fundamental component of modern clinical practice. Today, dental professionals are faced with a critical technological divergence: the choice between devices with Built-in AI (Edge Computing) and systems that rely on Cloud-Based AI processing. This document serves as a comprehensive technical comparison, analyzing the structural, functional, and economic implications of these two distinct architectural paradigms.
The “AI Built-in” model, often referred to as Edge AI, processes data directly on the dental device—be it an intraoral scanner, a milling machine, or a CBCT unit. This approach prioritizes immediacy, low latency, and operational independence from network connectivity. Conversely, the “Cloud-Based” model leverages the immense processing power of remote server farms to analyze high-definition (HD) photos, radiographs, and 3D scans. This approach prioritizes deep learning capabilities, massive dataset cross-referencing, and continuous algorithmic improvement.
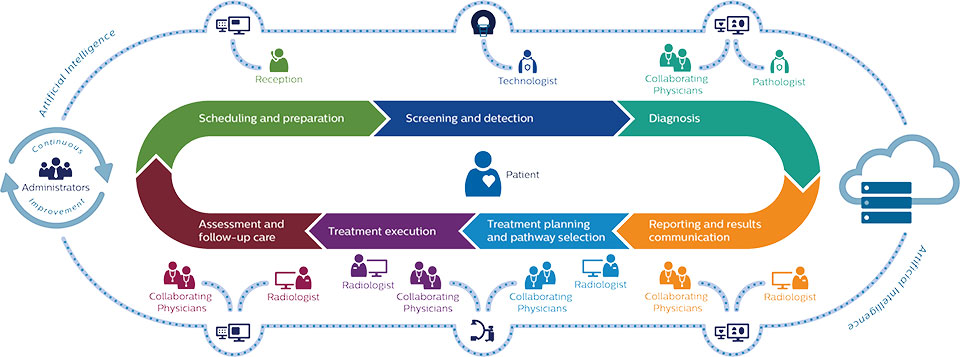
For the modern dental practice, this is not merely a hardware specification choice but a workflow decision. It affects how quickly a patient can be diagnosed chairside, how data is secured, the long-term cost of ownership, and the precision of complex treatment planning. As intraoral cameras and scanners now capture images in high definition (HD) and true color, the volume of data generated has exploded, making the efficiency of AI processing more critical than ever.
This report aims to demystify these technologies for decision-makers. We will explore how local processors handle real-time stitching of 3D models versus how cloud servers detect sub-clinical pathologies in panoramic X-rays. By the conclusion of this document, clinical directors and practitioners will possess the insights necessary to select the architecture that best aligns with their clinical philosophy and operational goals.
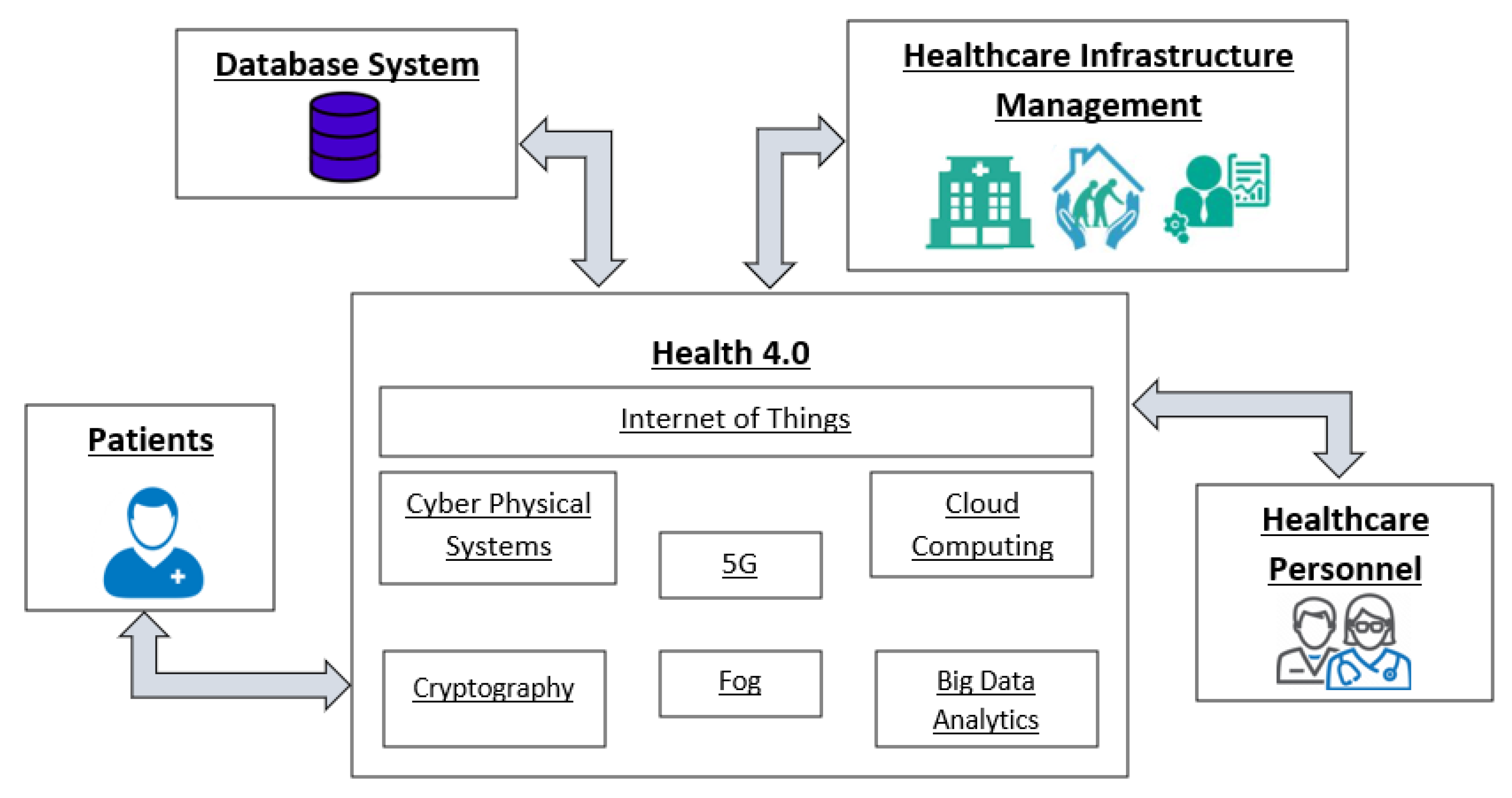
2. Understanding AI in Dental Technology
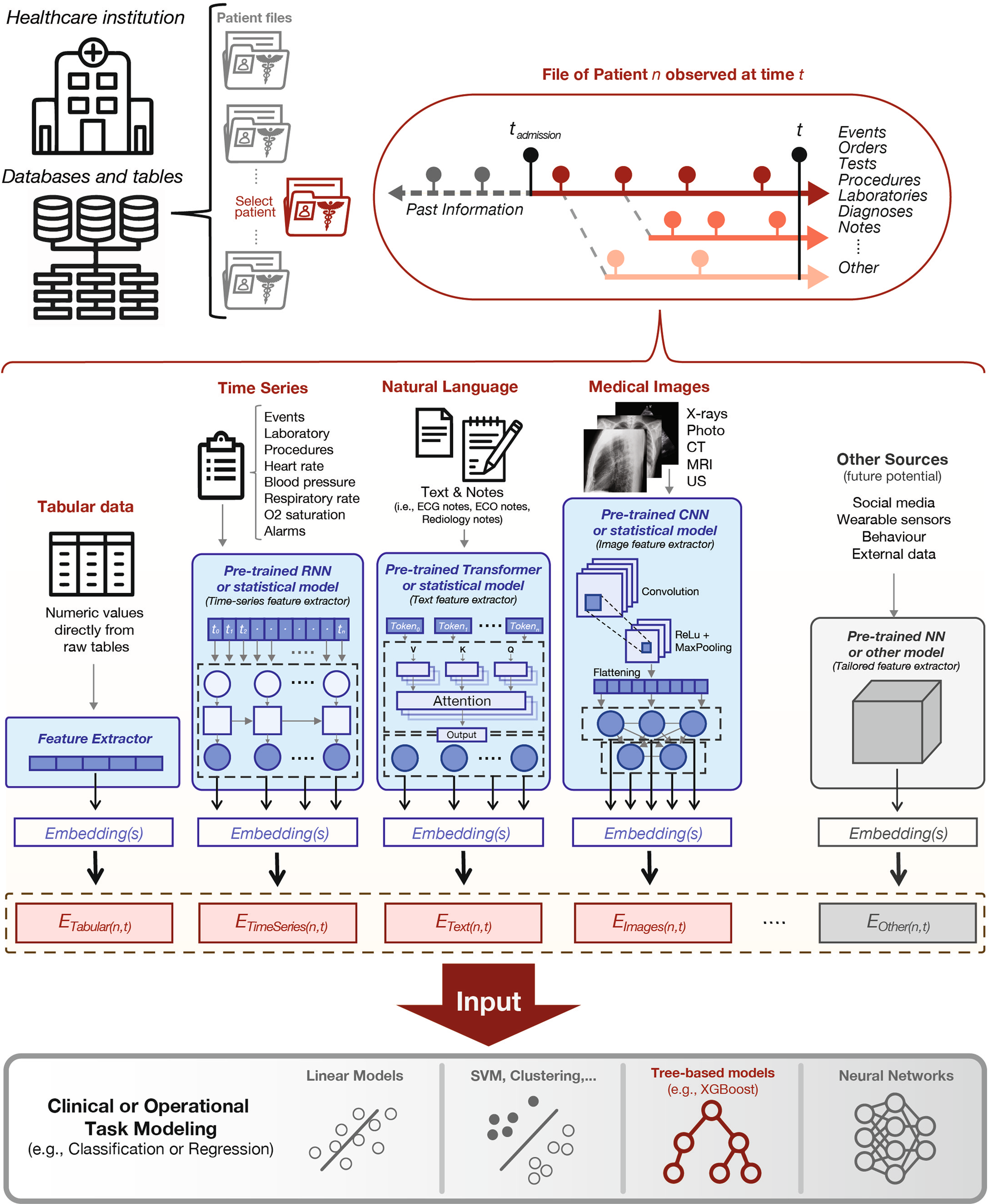
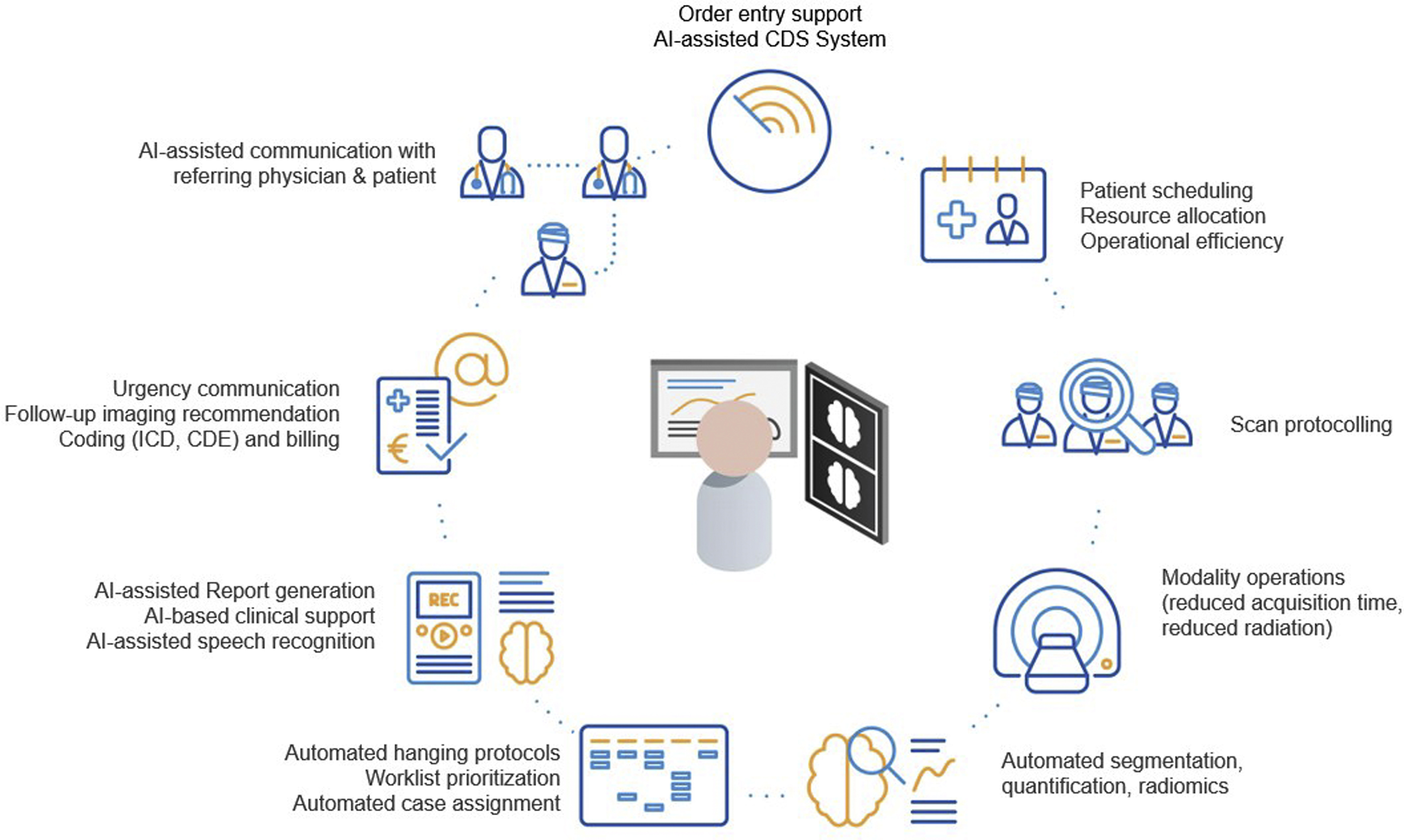
To evaluate the merits of built-in versus cloud-based systems, one must first understand the computational demands of dental AI. Artificial Intelligence in dentistry primarily utilizes Computer Vision—a field of AI that trains computers to interpret and understand the visual world. Using digital images from intraoral cameras, scanners, and radiographs, machines can now identify anatomical structures, detect pathologies like caries or bone loss, and propose treatment plans.
The Data Challenge: High Definition & 3D Volumes
Dental diagnostics involve heavy data loads. A single intraoral scan can generate millions of data points (point clouds) that must be stitched together in real-time to form a mesh. High-Definition (HD) intraoral photos require analysis of pixel-level color variations to detect early enamel demineralization. Processing this data requires significant computational power, usually in the form of Graphics Processing Units (GPUs).
Defining the Architectures
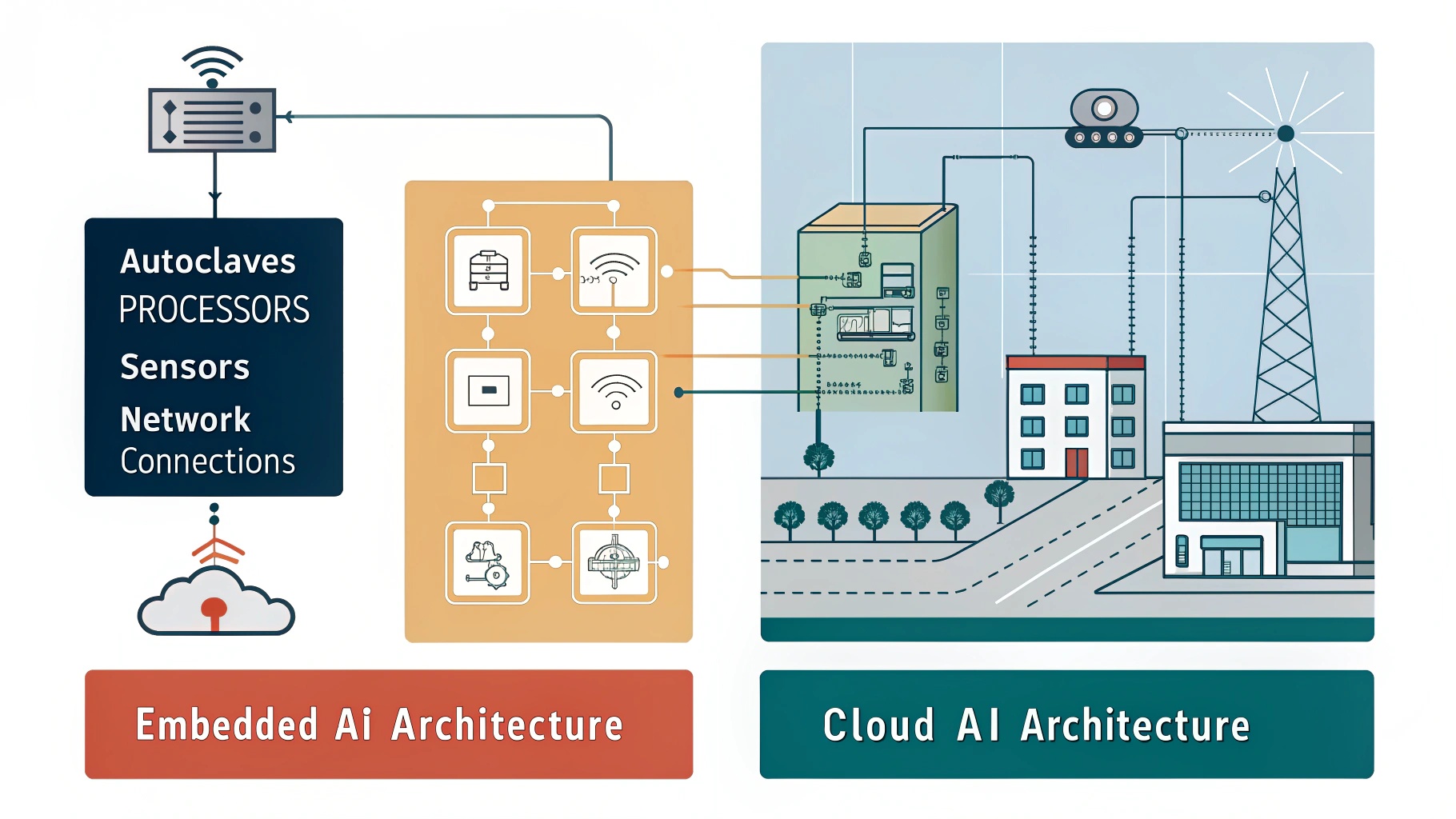
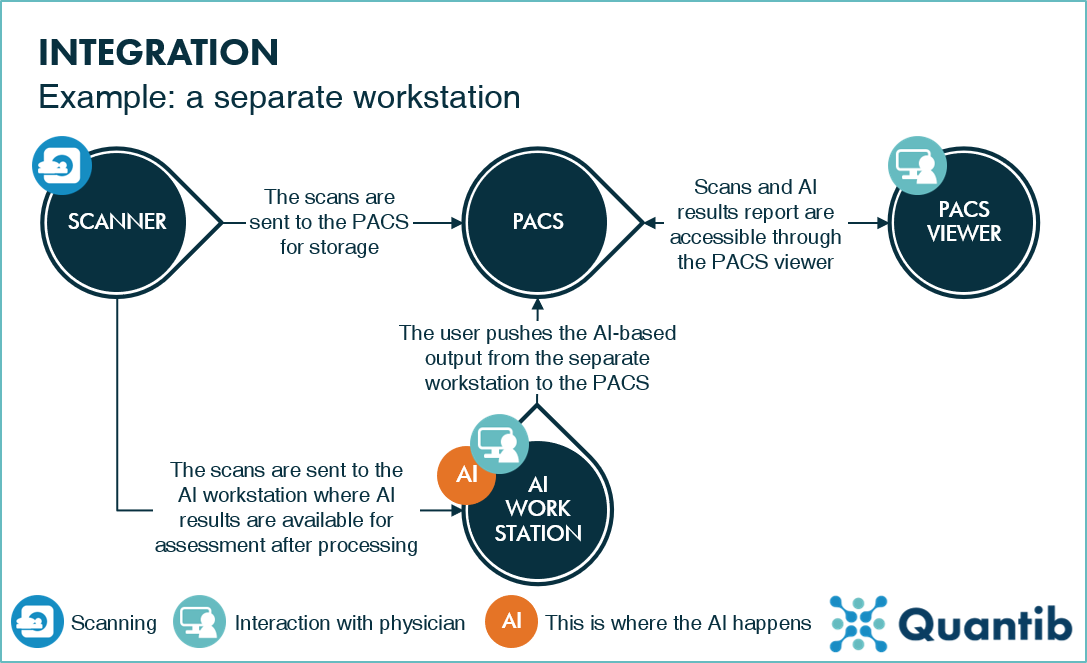
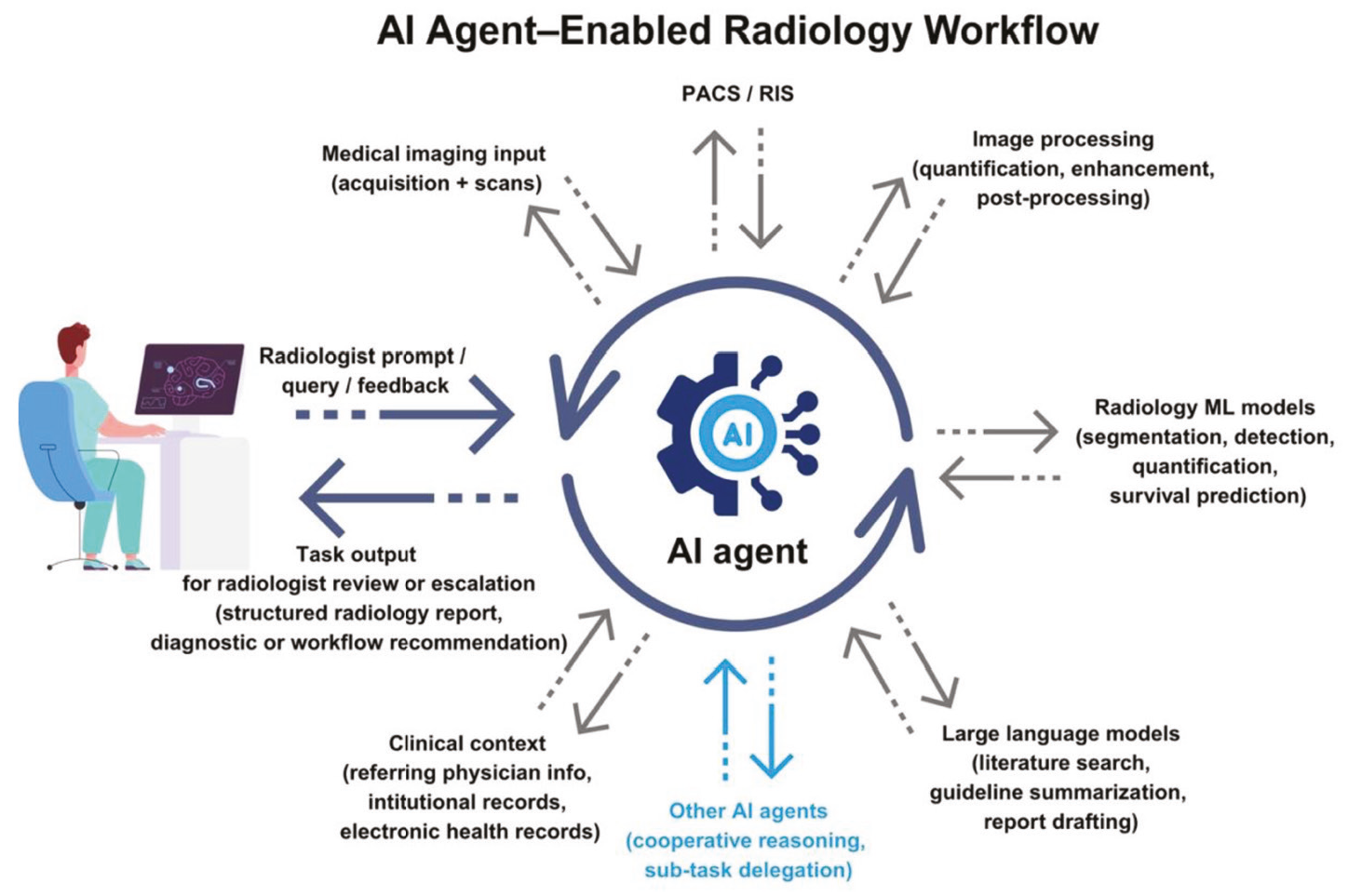
- Edge AI (Built-in): In this architecture, the AI model lives inside the medical device or the workstation computer connected directly to it. The “inference” (the act of the AI making a decision) happens locally. This is analogous to a reflex action in the human body—fast, localized, and instinctive.
- Cloud AI: In this architecture, the device is merely a data collection point. It captures the image or scan and transmits it securely via the internet to a centralized server (the cloud). The heavy processing happens remotely, and the results are sent back to the clinic. This is analogous to “thinking”—it requires more time and resources but allows for deeper contemplation and access to a broader base of knowledge.
The choice between these two architectures dictates the “User Experience” (UX) of the dentist. Does the scanner stop if the internet cuts out? Can the software detect a rare pathology that requires a database of millions of cases? These are the practical questions that stem from the architectural differences.
3. AI Built-in Dental Devices: Deep Dive
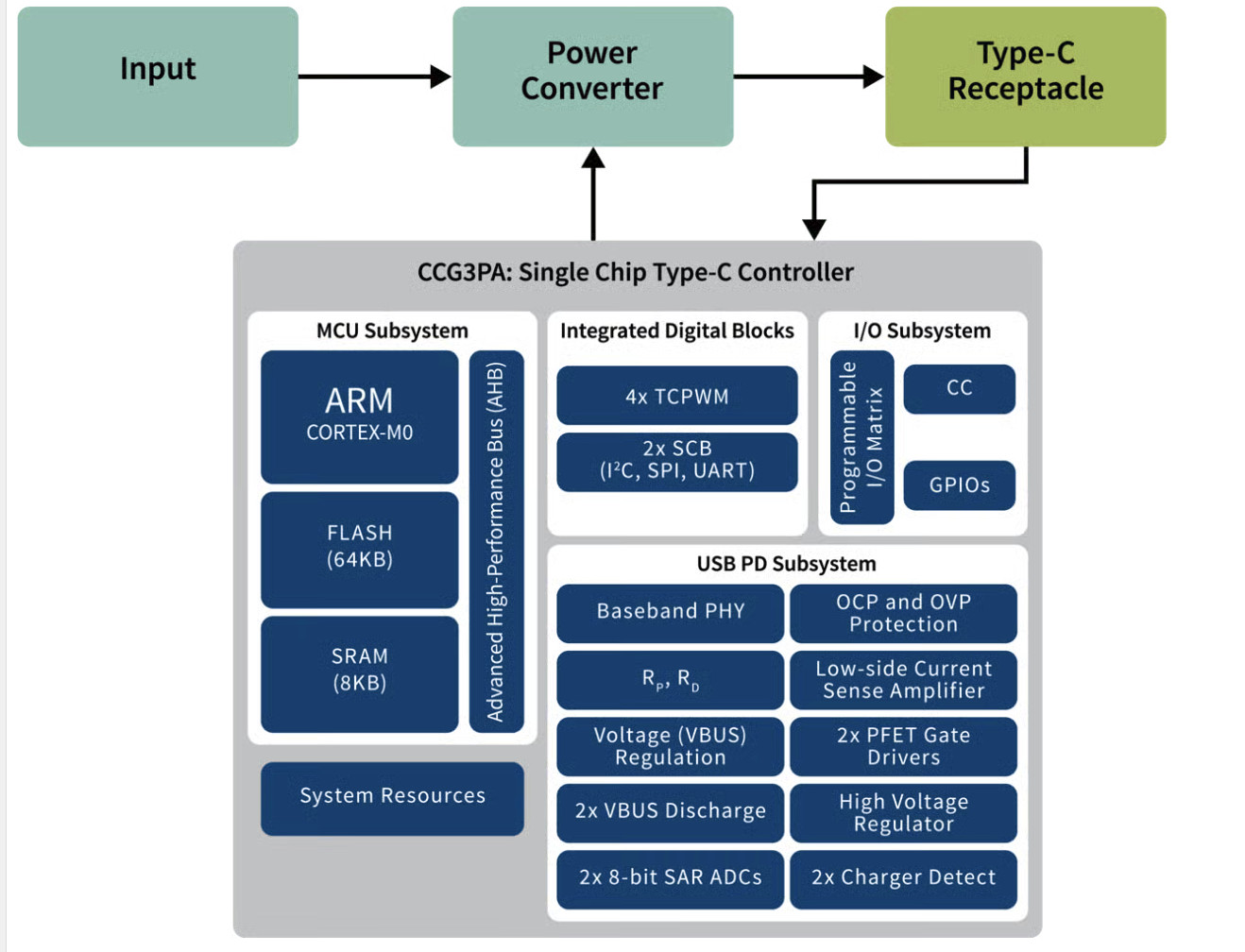
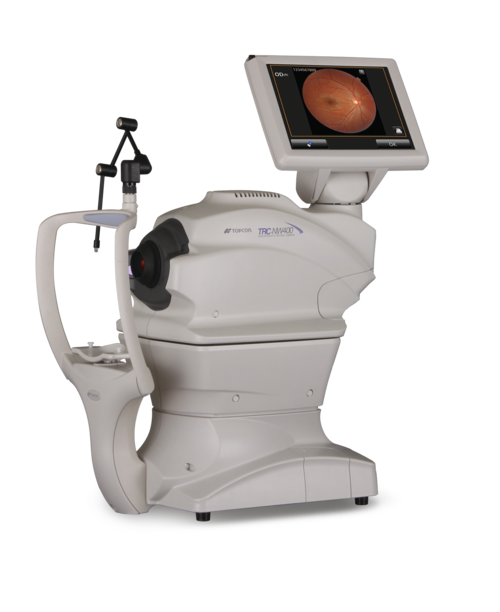
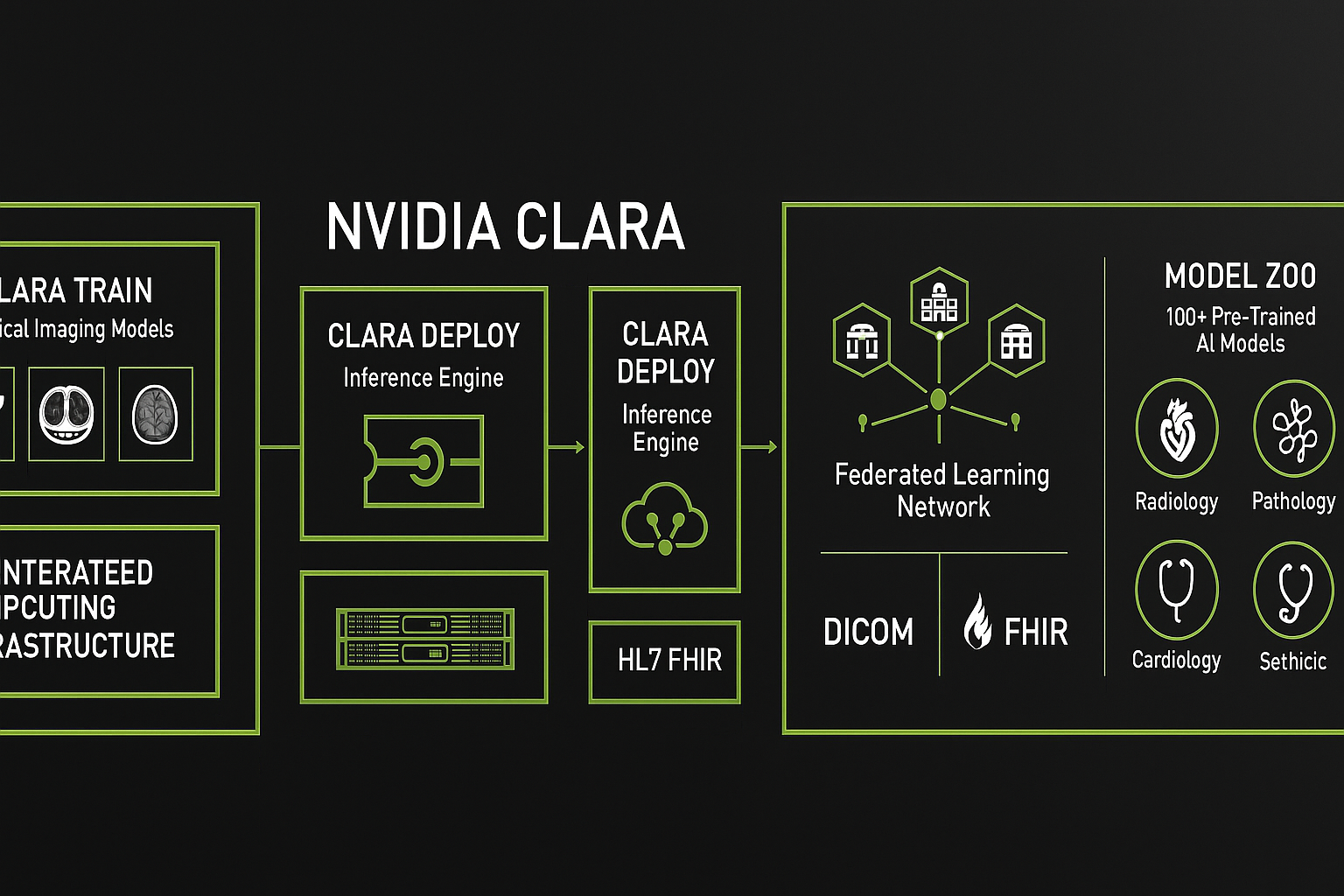
AI built-in devices represent the forefront of “Edge Computing” in healthcare. The primary characteristic of these devices is their autonomy. They are equipped with powerful internal processors (often high-end GPUs from manufacturers like NVIDIA) or specialized AI chips (TPUs/NPUs) that run algorithms natively.
Technology Overview
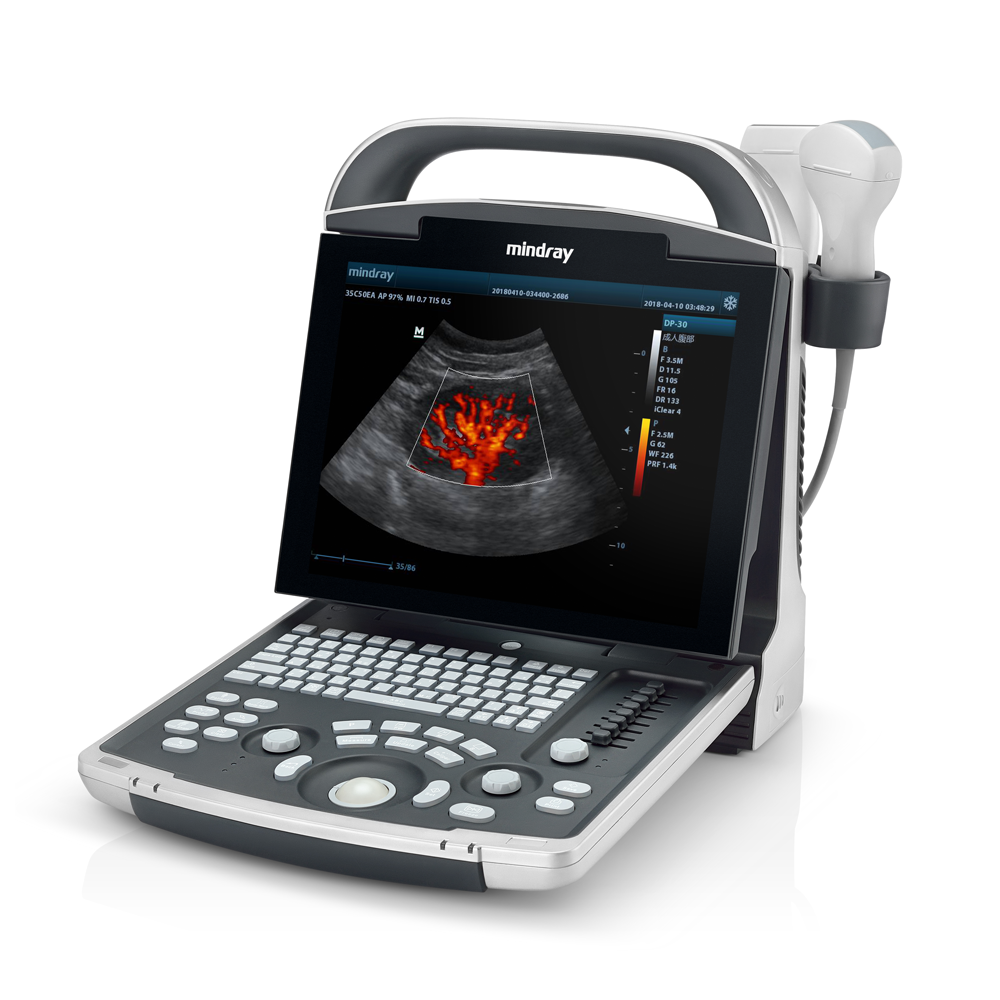
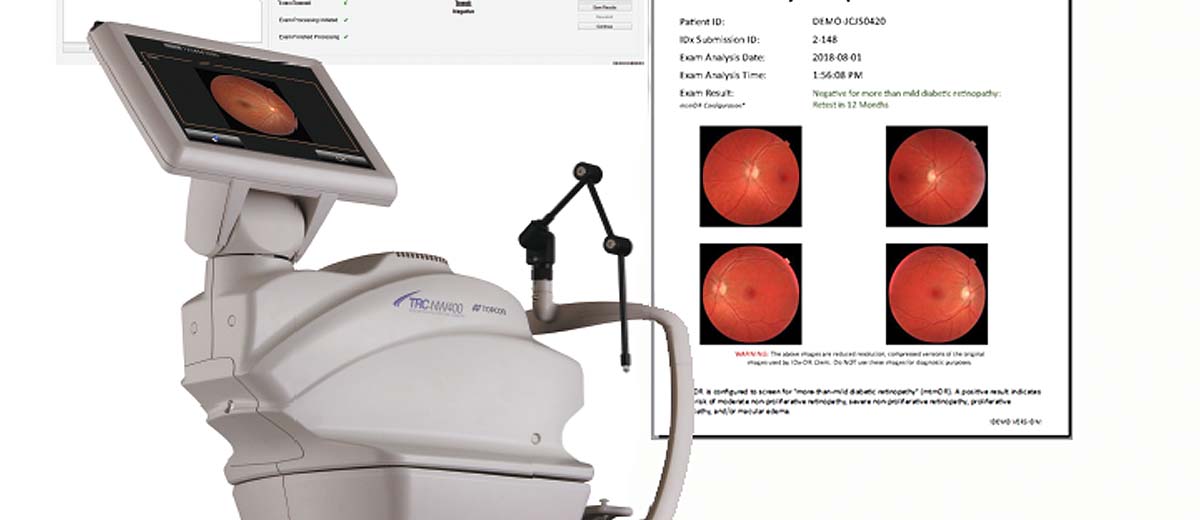
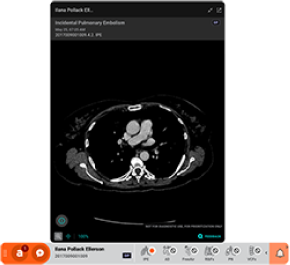
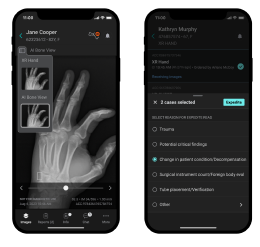
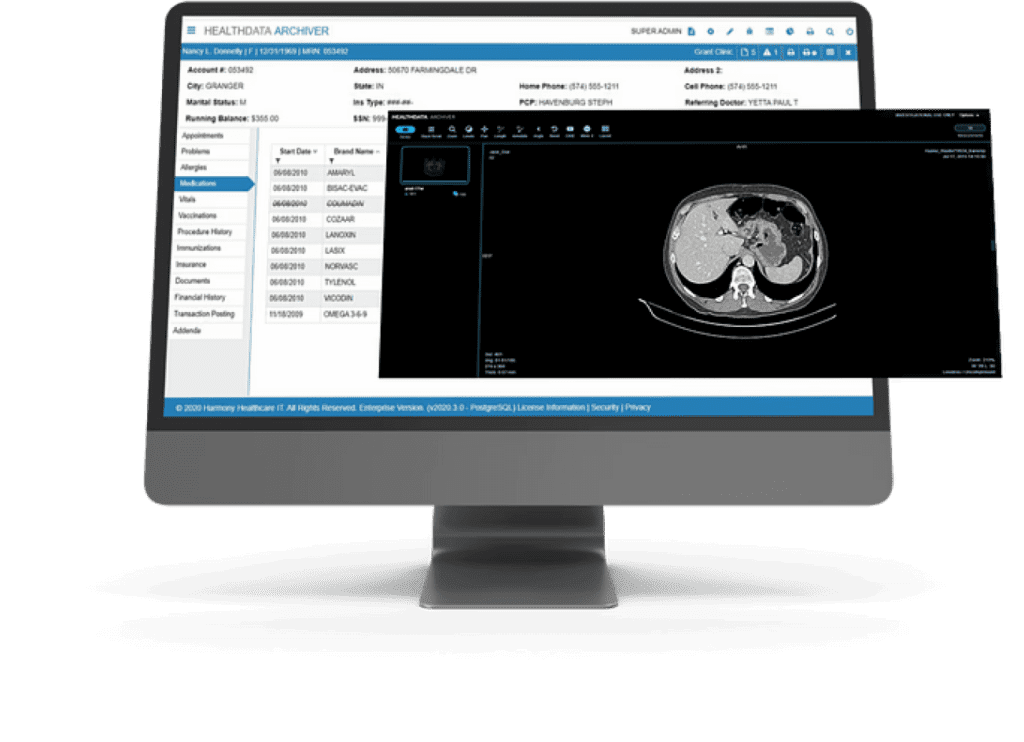
The most common example of AI built-in technology is the modern intraoral scanner (IOS). When a dentist scans a patient’s mouth, the scanner takes thousands of images per second. AI algorithms running locally on the system’s laptop or the wand itself must instantly recognize soft tissue (tongue, cheek) and filter it out, stitching only the hard tissue (teeth) into a 3D model. This “AI-driven scanning” must happen with zero latency; otherwise, the scanning experience would be jerky and unusable.
Key Features
- Real-Time Tissue Removal: As scan data enters the system, AI identifies the tongue, cheeks, and gloves, preventing them from being part of the final model.
- Instant Bite Registration: Built-in algorithms analyze the geometry of the upper and lower arches to automatically snap them into occlusion without manual alignment.
- Offline Functionality: Since the “brain” is local, these devices function perfectly without an internet connection, ensuring 100% uptime in clinics with unstable networks.
- Smart Guidance: Some devices offer visual cues (arrows or colors) on the screen in real-time, guiding the dentist to areas they missed during the scan.
Advantages
The paramount advantage of built-in AI is speed. In procedures like digital impressions, a delay of even 500 milliseconds between the hand movement and the screen update is perceptible and frustrating. Built-in AI eliminates network latency. Furthermore, data privacy is inherently easier to manage in strict environments, as the raw data does not necessarily need to leave the premises to be processed.
Limitations
The limitation of this architecture is hardware dependency. To run advanced AI, the dental office must purchase expensive computers with high-end graphics cards. The “intelligence” of the device is also “frozen” at the time of the software update; it does not learn continuously from global data unless a firmware update is manually installed. Additionally, the complexity of the AI models is limited by the local hardware’s power—it cannot run the massive neural networks that a cloud server can.
4. Cloud-Based AI Dental Devices: Deep Dive
Cloud-based AI represents the “Big Data” approach to dentistry. In this model, the clinical device acts primarily as a high-definition capture terminal. The heavy lifting of diagnosis, segmentation, and treatment planning occurs on remote servers complying with HIPAA/GDPR standards.
Technology Overview
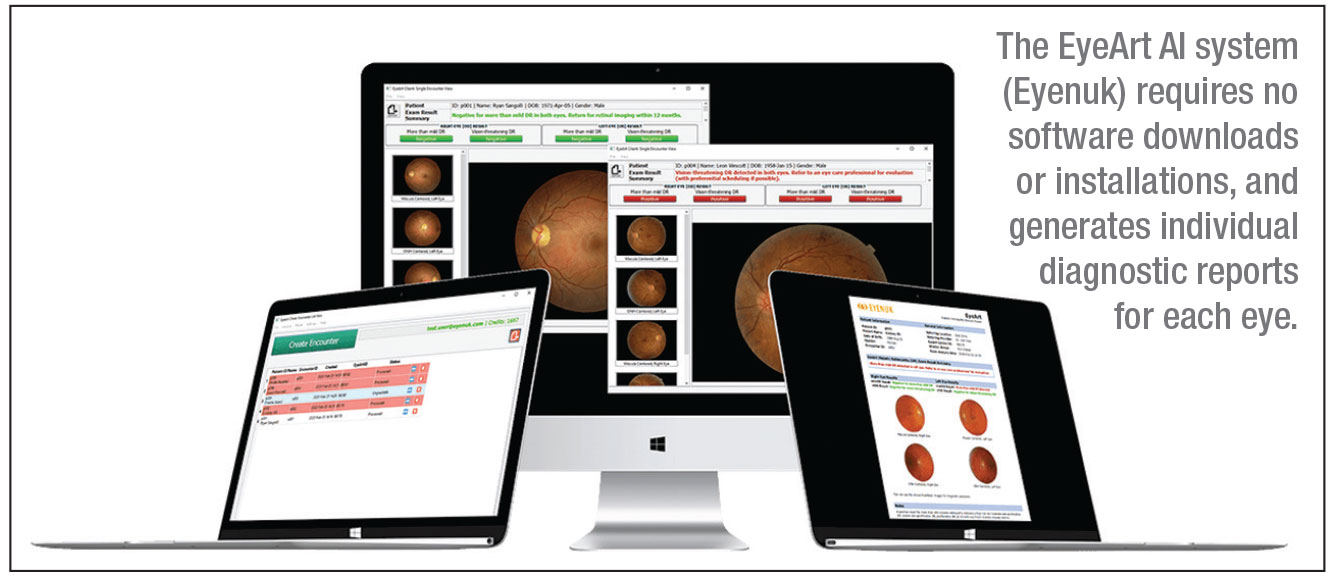
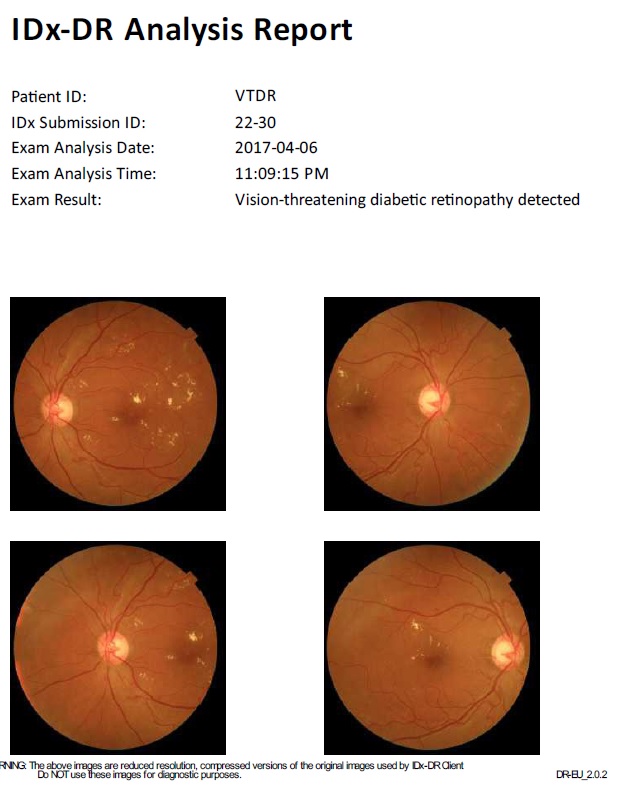
This ecosystem is exemplified by radiographic analysis platforms and CAD/CAM design services. A dentist takes a set of HD bitewings or a CBCT scan. This large dataset is uploaded to the cloud. There, enterprise-grade GPUs run incredibly complex Convolutional Neural Networks (CNNs) that have been trained on millions of annotated images. Within seconds or minutes, the system returns a fully annotated report highlighting caries, apical lucencies, and bone levels.
Key Features
- Deep Diagnostic Analysis: Unlike the “reflex” AI of built-in scanners, cloud AI provides “reflective” analysis, identifying subtle pathologies that require massive reference datasets.
- Continuous Learning: Cloud models update constantly. Every confirmed diagnosis adds to the global dataset, making the AI smarter for every user instantly without software installations.
- Hardware Agnosticism: Because the processing happens remotely, the dentist does not need a $5,000 gaming laptop. A basic tablet or office PC with a browser is sufficient to view the results.
- Interoperability: Cloud AI can easily integrate data from different sources (intraoral photos, X-rays, periodontal charts) to create a holistic patient view.
Advantages
The major benefit is diagnostic depth and scalability. Cloud systems can afford to run algorithms that would melt a laptop processor. They also offer a lower barrier to entry regarding hardware costs. Furthermore, for multi-location practices (DSOs), cloud AI centralizes data, allowing clinical directors to audit diagnosis quality across all branches remotely.
Limitations
The Achilles’ heel of cloud AI is latency and connectivity. If the internet goes down, the diagnostic capability is lost. Uploading large 3D datasets (like CBCTs) can take time depending on bandwidth. There is also the perennial concern of data sovereignty—some institutions are legally restricted from uploading patient health information to third-party cloud servers.
5. Comparative Analysis
To assist in the decision-making process, we present a direct comparison across five critical dimensions: Performance, Cost, Security, Scalability, and Use Cases.
Critical Insight: The Latency vs. Accuracy Trade-offBuilt-in AI wins on Latency (immediate response for motor tasks like scanning). Cloud AI wins on Accuracy for complex diagnostic tasks (identifying pathologies in static images).
Performance Comparison
Performance must be defined by the task. For procedural tasks (scanning, milling), built-in AI is superior. The feedback loop must be instant. For diagnostic tasks (reading X-rays, treatment planning), cloud AI is superior because accuracy is more important than speed, and the computational complexity requires server-grade hardware.
Cost Analysis
| Factor | Built-in AI Devices | Cloud-Based AI Systems |
|---|---|---|
| Upfront Cost | High (Requires powerful hardware/GPUs) | Low (Uses existing office PCs/Tablets) |
| Recurring Cost | Low (Usually included in purchase or maintenance) | High (SaaS subscription models) |
| Depreciation | Hardware becomes obsolete in 3-5 years | Hardware stays relevant; server upgrades happen remotely |
Security and Privacy
Built-in AI keeps data essentially within the clinic’s Local Area Network (LAN), which appeals to privacy-conscious practices. However, local data is susceptible to ransomware attacks if not backed up. Cloud AI providers invest millions in enterprise-grade security (encryption at rest and in transit), often exceeding what a local IT provider can offer. However, the transmission of data inherently introduces a theoretical interception risk, mitigated by strict TLS protocols.
Use Case Scenarios
- Choose Built-in AI if: You are a solo practitioner in a rural area with poor internet; you prioritize chairside speed for impressions; you dislike subscription fees.
- Choose Cloud AI if: You are a DSO looking for standardized diagnosis; you want the highest accuracy in pathology detection; you prefer lower upfront capital expenditure (CapEx) and operating expenditure (OpEx).
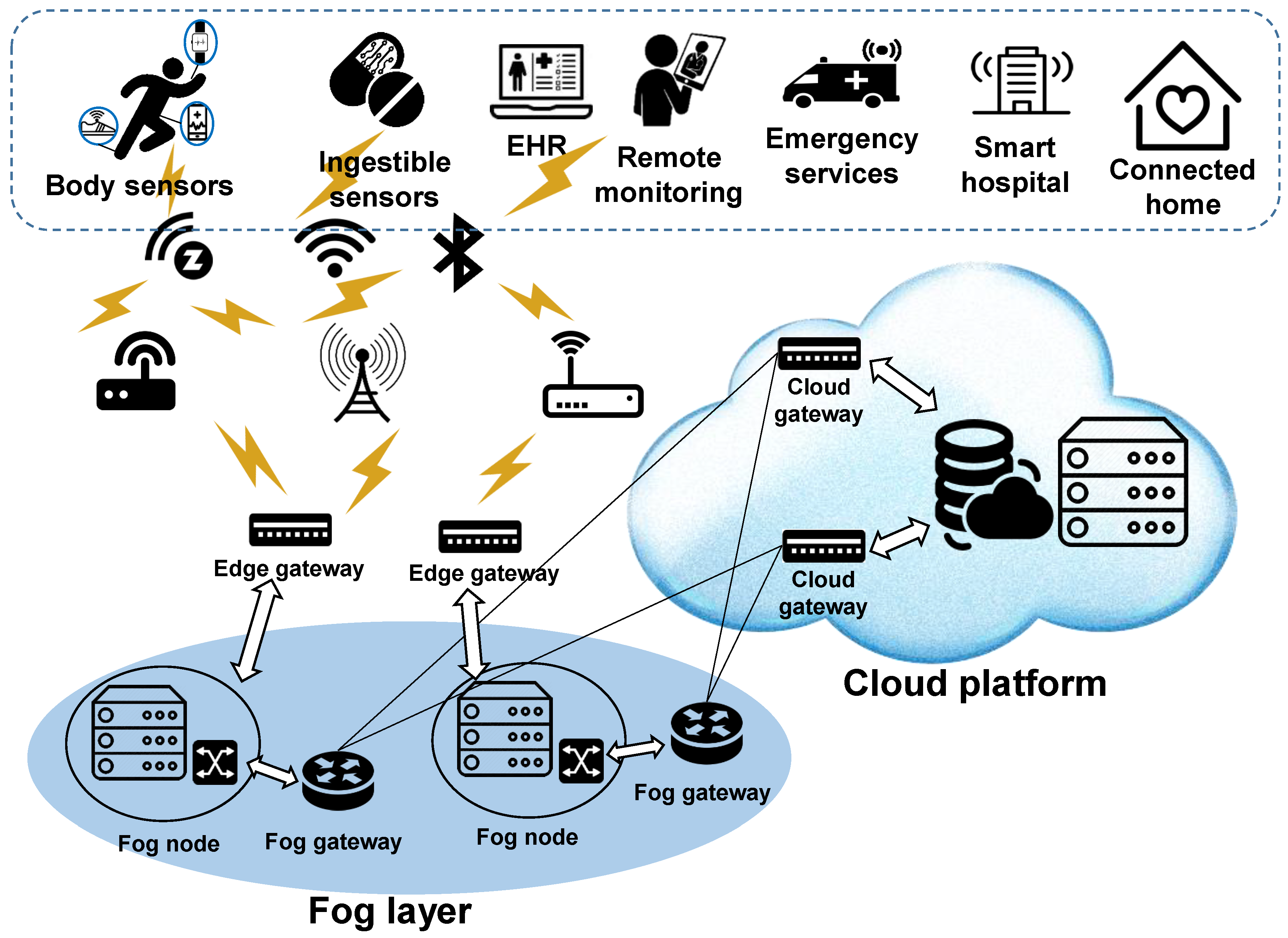
6. Future Trends and Hybrid Solutions
The strict dichotomy between Edge and Cloud is beginning to blur, giving rise to Hybrid AI or “Fog Computing.” In this emerging model, devices perform lightweight AI tasks locally (like tissue filtration during scanning) while simultaneously uploading data to the cloud for heavy-duty analysis (like final model optimization and caries check).
The Impact of 5G: As 5G networks become ubiquitous, the latency issues of cloud computing will diminish. This may allow “dumber” scanning wands to stream raw video directly to the cloud for real-time processing, reducing the size and cost of the physical device in the dentist’s hand.
Federated Learning: This is a privacy-preserving future trend where the Built-in AI on your office device learns from your local data and sends only the learnings (not the patient images) to the cloud global model. This combines the privacy of Edge AI with the collective intelligence of Cloud AI.
7. Conclusion and Recommendations
The choice between AI built-in and cloud-based dental devices is not a binary one of “better” or “worse,” but rather a strategic alignment with clinical workflow requirements.
For Imaging and Acquisition: We recommend Built-in AI. The requirement for real-time motor feedback during intraoral scanning necessitates the zero-latency environment that only local processing can provide. A scanner that relies on the cloud for basic stitching is currently impractical.
For Diagnostics and Planning: We recommend Cloud-Based AI. The ability to leverage massive, constantly updated neural networks provides a level of diagnostic assurance—a “second opinion”—that local hardware cannot match. The subscription cost is offset by the increased case acceptance and diagnostic accuracy.
Ultimately, the modern digital dental clinic will likely be a hybrid environment. It will feature powerful edge devices for data capture that seamlessly feed into cloud ecosystems for analysis, storage, and design. Practitioners investing in technology today should ensure their hardware vendors offer open APIs and cloud connectivity to future-proof their practice against this inevitable convergence.
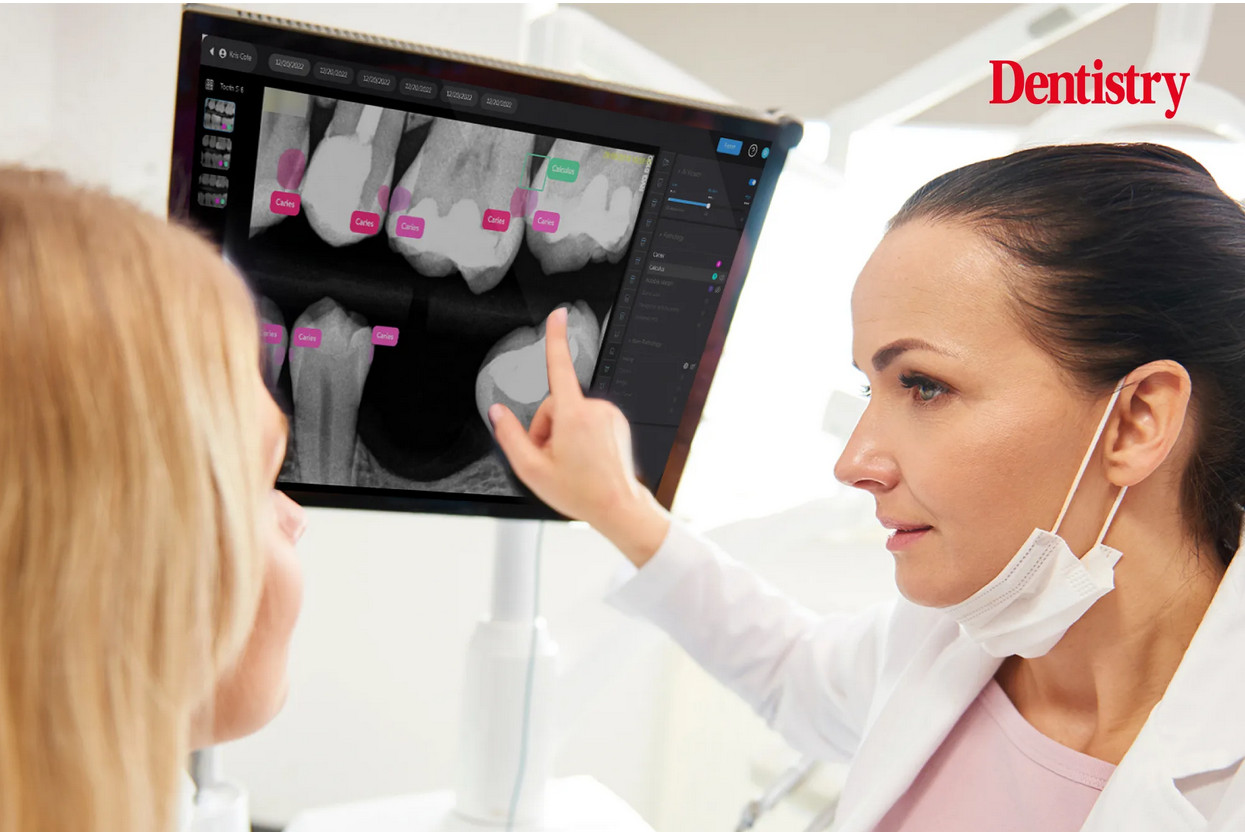




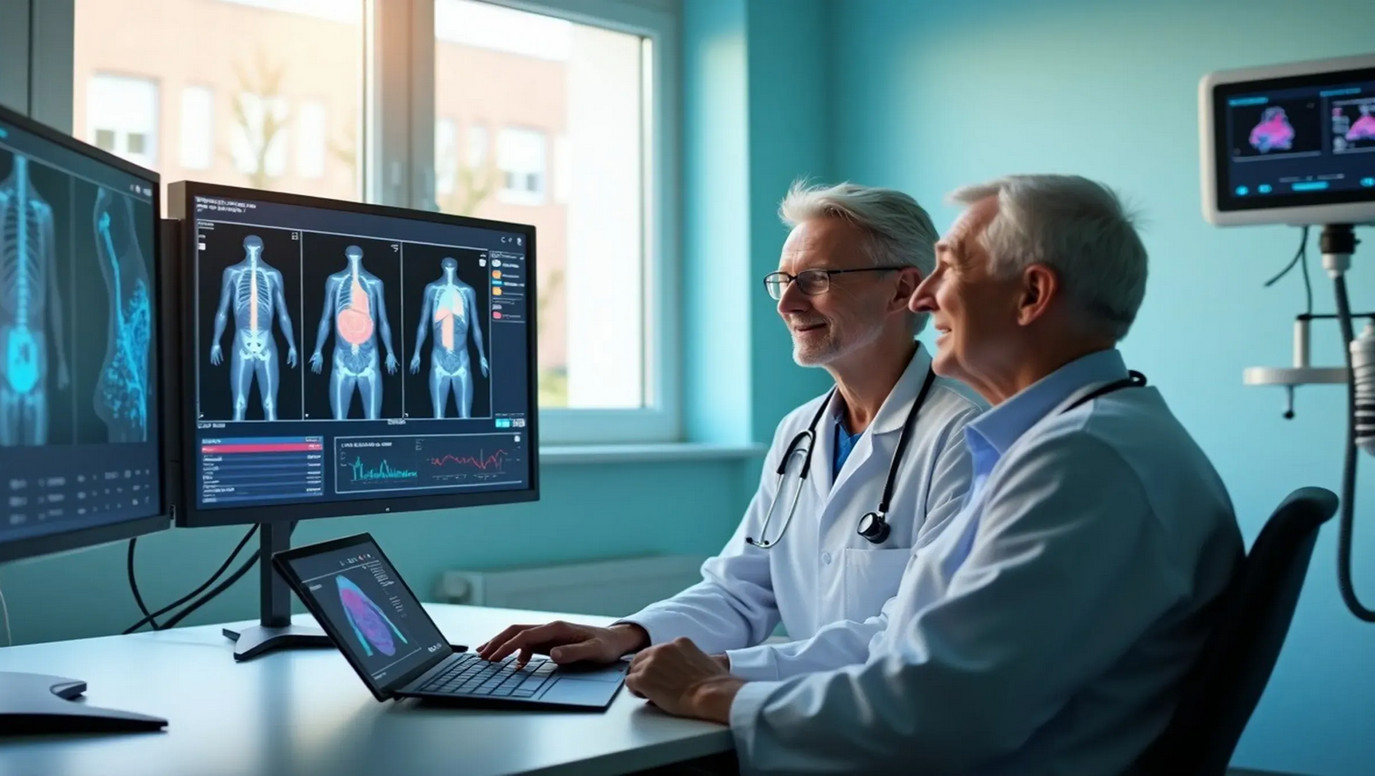











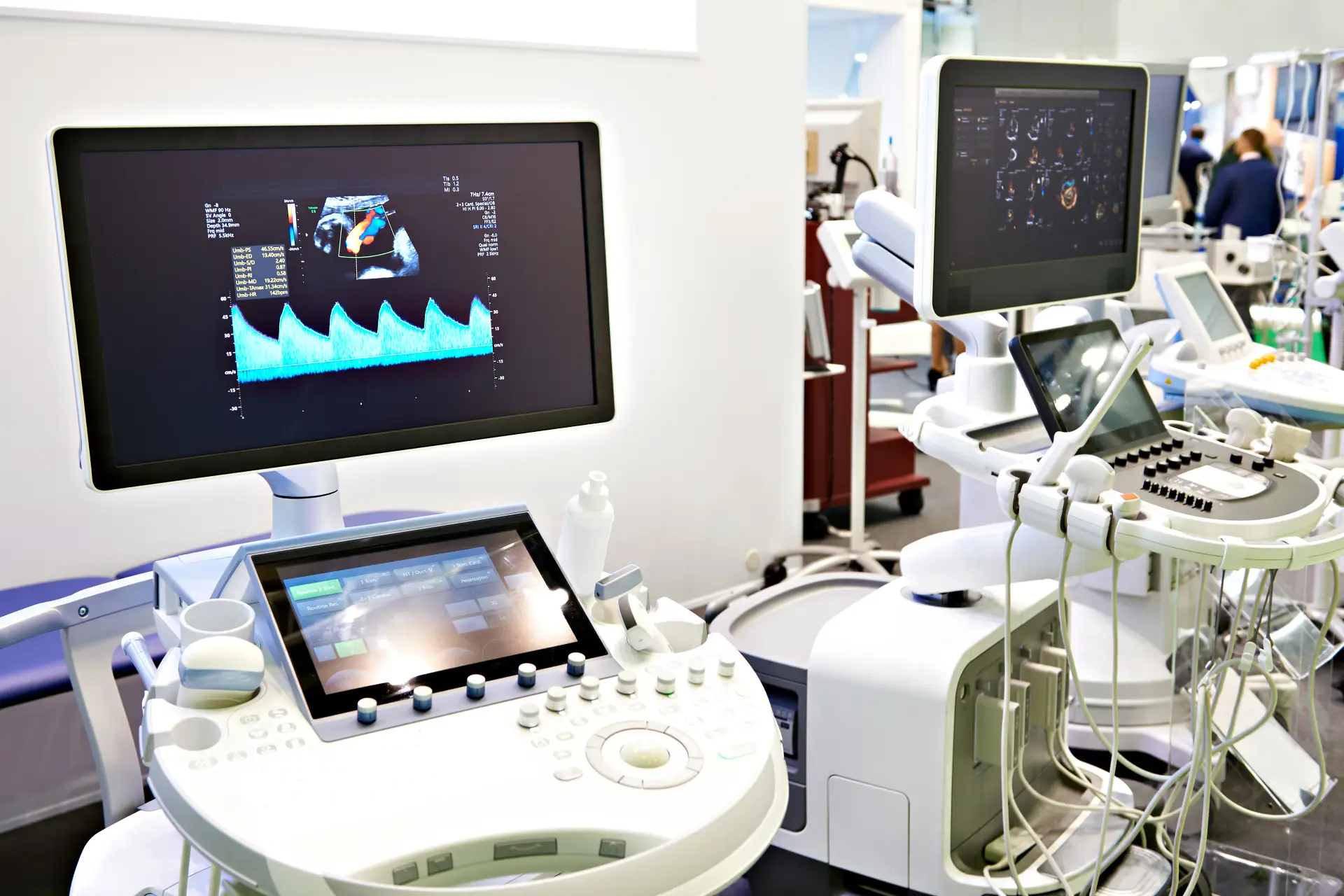
 Heart disease, specifically ischemic heart disease, remains the leading cause of death in the United States, responsible for nearly one-quarter of all fatalities. The electrocardiograph (ECG or EKG) machine is the cornerstone device for detecting cardiac abnormalities. These sophisticated instruments record the electrical activity of the heart over time, identifying arrhythmias, myocardial infarctions (heart attacks), atrial fibrillation, and ventricular tachycardia. Modern ECG machines feature USB connectivity, PC software integration, and often include 12-lead capabilities for comprehensive cardiac assessment. Portable versions like the CONTEC ECG600G enable continuous monitoring, while hospital-grade systems provide real-time analysis and automated diagnostic suggestions. The technology has evolved to include wireless transmission, cloud storage, and AI-powered interpretation, making early detection more accessible and accurate.
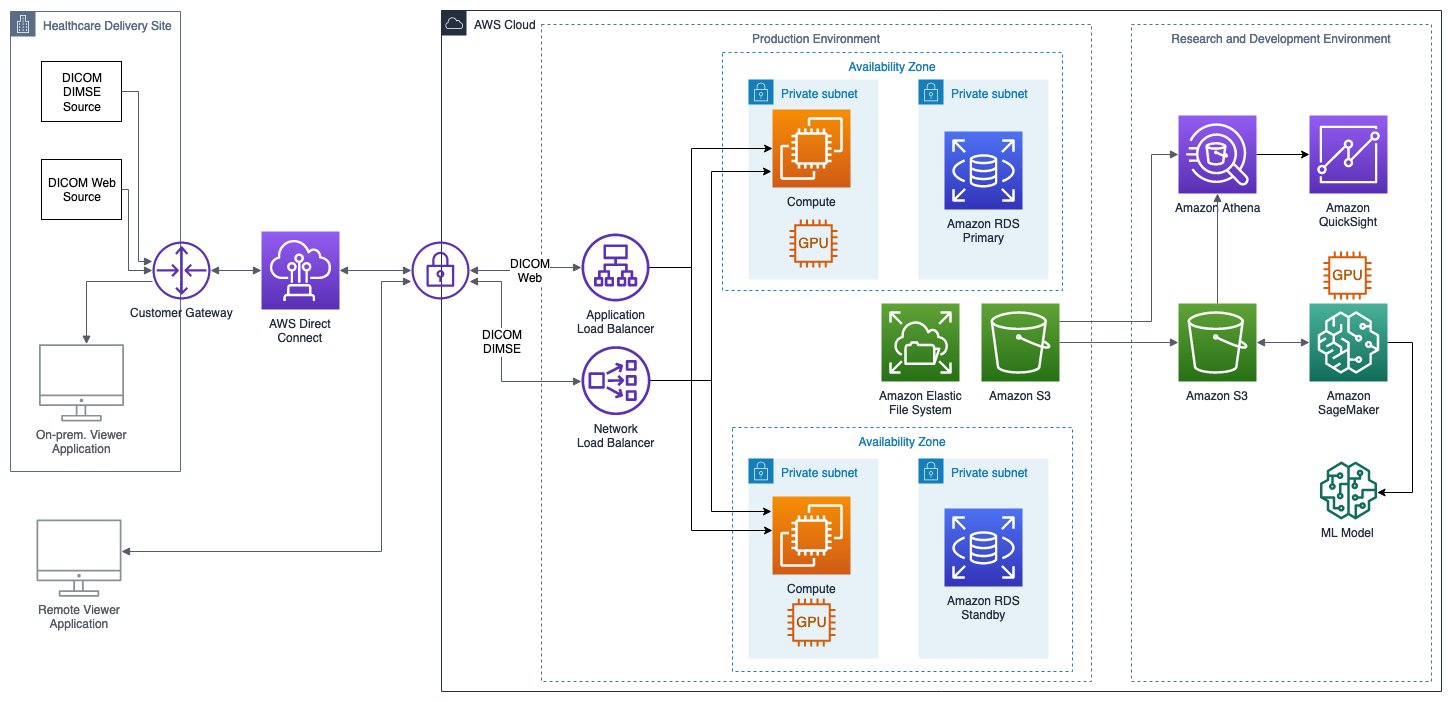
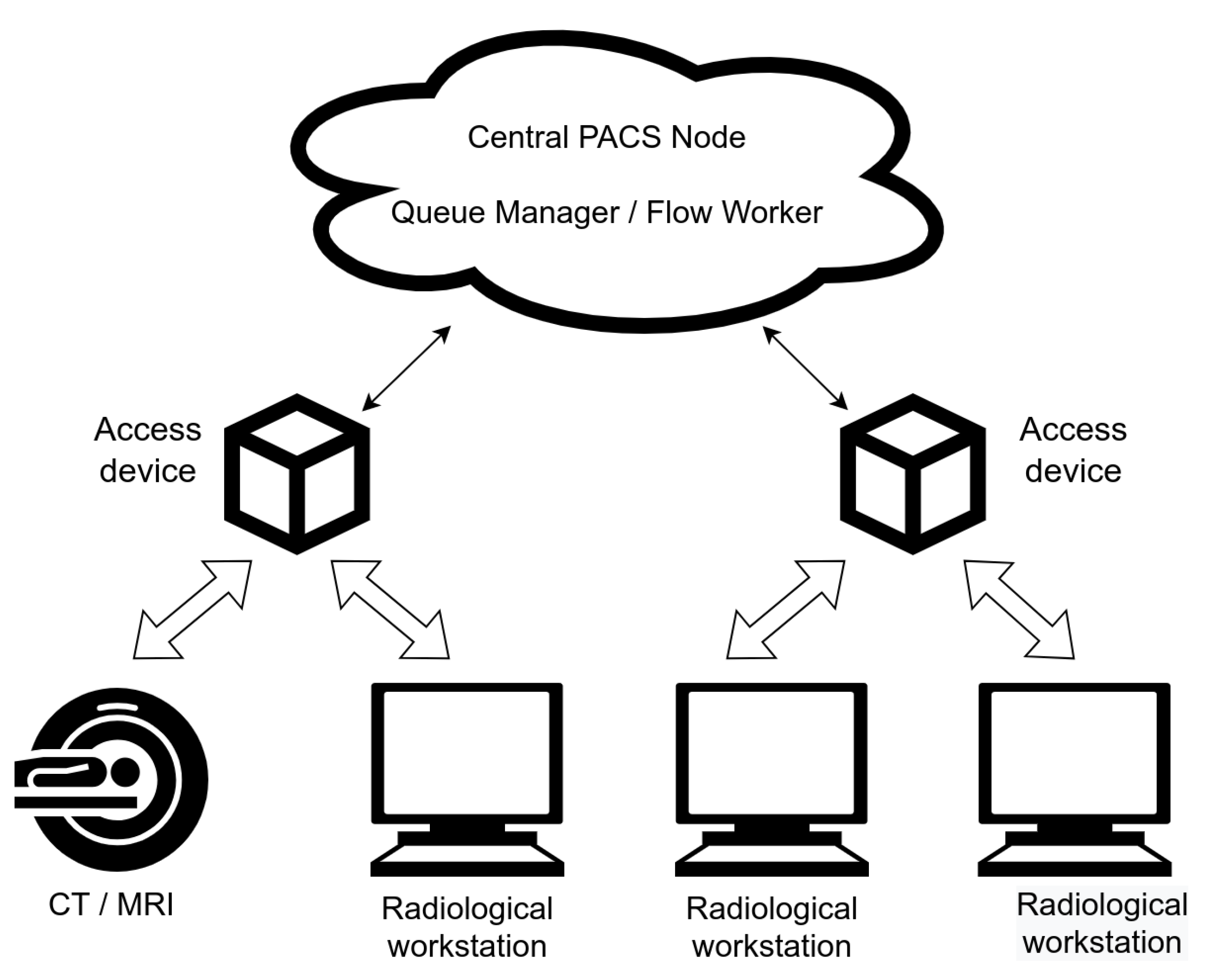
Heart disease, specifically ischemic heart disease, remains the leading cause of death in the United States, responsible for nearly one-quarter of all fatalities. The electrocardiograph (ECG or EKG) machine is the cornerstone device for detecting cardiac abnormalities. These sophisticated instruments record the electrical activity of the heart over time, identifying arrhythmias, myocardial infarctions (heart attacks), atrial fibrillation, and ventricular tachycardia. Modern ECG machines feature USB connectivity, PC software integration, and often include 12-lead capabilities for comprehensive cardiac assessment. Portable versions like the CONTEC ECG600G enable continuous monitoring, while hospital-grade systems provide real-time analysis and automated diagnostic suggestions. The technology has evolved to include wireless transmission, cloud storage, and AI-powered interpretation, making early detection more accessible and accurate. Malignant neoplasms (cancer) represent the second leading cause of death, affecting over 619,000 Americans annually. The primary detection equipment includes Computed Tomography (CT) scanners and Magnetic Resonance Imaging (MRI) systems. CT scanners use X-ray technology to create detailed cross-sectional images of the body, excelling at detecting lung cancer, colorectal cancer, and metastatic disease. Modern multi-slice CT scanners can generate thousands of images in minutes, with specialized protocols for low-dose lung cancer screening. MRI systems utilize powerful magnetic fields and radio waves to produce high-resolution images, particularly effective for brain tumors, breast cancer, and soft tissue malignancies. Advanced systems now incorporate PET-CT fusion technology, combining metabolic and anatomical imaging for superior diagnostic accuracy. These systems connect to hospital PACS (Picture Archiving and Communication Systems) via USB and network interfaces, enabling rapid image sharing and telemedicine consultations.
Malignant neoplasms (cancer) represent the second leading cause of death, affecting over 619,000 Americans annually. The primary detection equipment includes Computed Tomography (CT) scanners and Magnetic Resonance Imaging (MRI) systems. CT scanners use X-ray technology to create detailed cross-sectional images of the body, excelling at detecting lung cancer, colorectal cancer, and metastatic disease. Modern multi-slice CT scanners can generate thousands of images in minutes, with specialized protocols for low-dose lung cancer screening. MRI systems utilize powerful magnetic fields and radio waves to produce high-resolution images, particularly effective for brain tumors, breast cancer, and soft tissue malignancies. Advanced systems now incorporate PET-CT fusion technology, combining metabolic and anatomical imaging for superior diagnostic accuracy. These systems connect to hospital PACS (Picture Archiving and Communication Systems) via USB and network interfaces, enabling rapid image sharing and telemedicine consultations. Cerebrovascular disease, particularly ischemic and hemorrhagic strokes, claims over 162,000 American lives annually. Emergency stroke detection relies on rapid brain imaging using CT and MRI scanners. Non-contrast CT scans can distinguish between ischemic stroke (caused by blood clots) and hemorrhagic stroke (caused by bleeding) within minutes, critical for determining treatment protocols. Advanced diffusion-weighted MRI (DW-MRI) can detect ischemic changes within minutes of onset, even before they appear on CT scans. Modern stroke centers employ mobile stroke units equipped with portable CT scanners, bringing diagnostic capabilities directly to patients, significantly reducing time-to-treatment.
Cerebrovascular disease, particularly ischemic and hemorrhagic strokes, claims over 162,000 American lives annually. Emergency stroke detection relies on rapid brain imaging using CT and MRI scanners. Non-contrast CT scans can distinguish between ischemic stroke (caused by blood clots) and hemorrhagic stroke (caused by bleeding) within minutes, critical for determining treatment protocols. Advanced diffusion-weighted MRI (DW-MRI) can detect ischemic changes within minutes of onset, even before they appear on CT scans. Modern stroke centers employ mobile stroke units equipped with portable CT scanners, bringing diagnostic capabilities directly to patients, significantly reducing time-to-treatment. Chronic obstructive pulmonary disease (COPD) and related respiratory conditions affect millions of Americans. The spirometer is the gold standard for detecting and monitoring these conditions. This device measures lung function by assessing the volume and speed of air that can be inhaled and exhaled. Modern digital spirometers, such as the SP100A and JYTOP SP80B models, feature USB connectivity and PC software that generates comprehensive reports including FEV1 (Forced Expiratory Volume), FVC (Forced Vital Capacity), and peak expiratory flow rate. These portable devices enable both clinical diagnosis and home monitoring, with color displays showing real-time waveforms and comparing results against predicted values based on age, height, and gender.
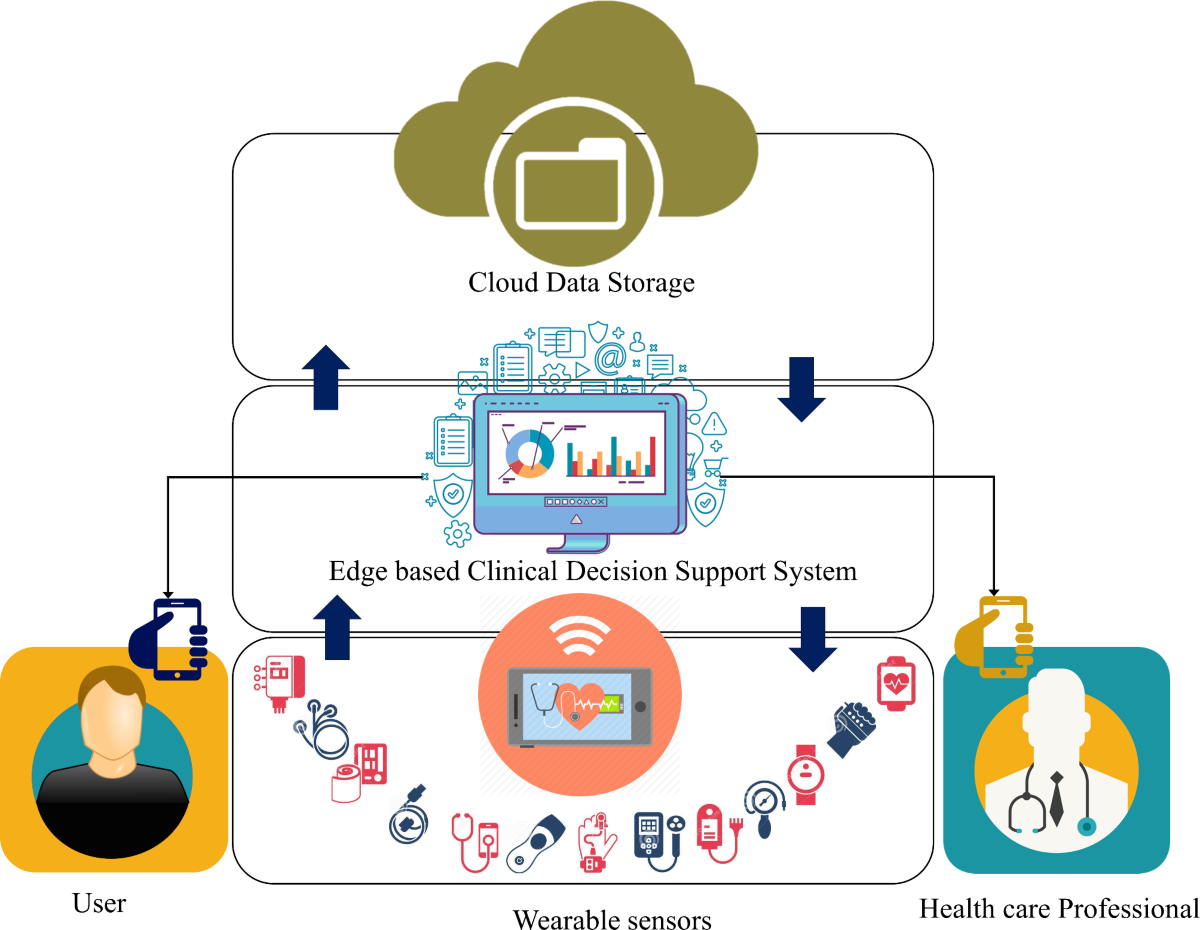
Chronic obstructive pulmonary disease (COPD) and related respiratory conditions affect millions of Americans. The spirometer is the gold standard for detecting and monitoring these conditions. This device measures lung function by assessing the volume and speed of air that can be inhaled and exhaled. Modern digital spirometers, such as the SP100A and JYTOP SP80B models, feature USB connectivity and PC software that generates comprehensive reports including FEV1 (Forced Expiratory Volume), FVC (Forced Vital Capacity), and peak expiratory flow rate. These portable devices enable both clinical diagnosis and home monitoring, with color displays showing real-time waveforms and comparing results against predicted values based on age, height, and gender. Diabetes affects over 38 million Americans, with Type 2 diabetes being the most prevalent form. Blood glucose meters are essential for diabetes management and monitoring. Modern glucometers like those with USB data cables (FEANTEEK, I-sens 710) allow patients to track blood sugar levels and transmit data to healthcare providers. Advanced continuous glucose monitoring (CGM) systems provide real-time glucose readings throughout the day and night, alerting users to dangerous highs or lows. These devices connect via USB or wireless protocols to smartphones and computers, enabling comprehensive data analysis and trend identification.
Diabetes affects over 38 million Americans, with Type 2 diabetes being the most prevalent form. Blood glucose meters are essential for diabetes management and monitoring. Modern glucometers like those with USB data cables (FEANTEEK, I-sens 710) allow patients to track blood sugar levels and transmit data to healthcare providers. Advanced continuous glucose monitoring (CGM) systems provide real-time glucose readings throughout the day and night, alerting users to dangerous highs or lows. These devices connect via USB or wireless protocols to smartphones and computers, enabling comprehensive data analysis and trend identification.













.png?width=865&name=Pillar%20page%20-%20figure%2012-compressed2-min%20(1).png)




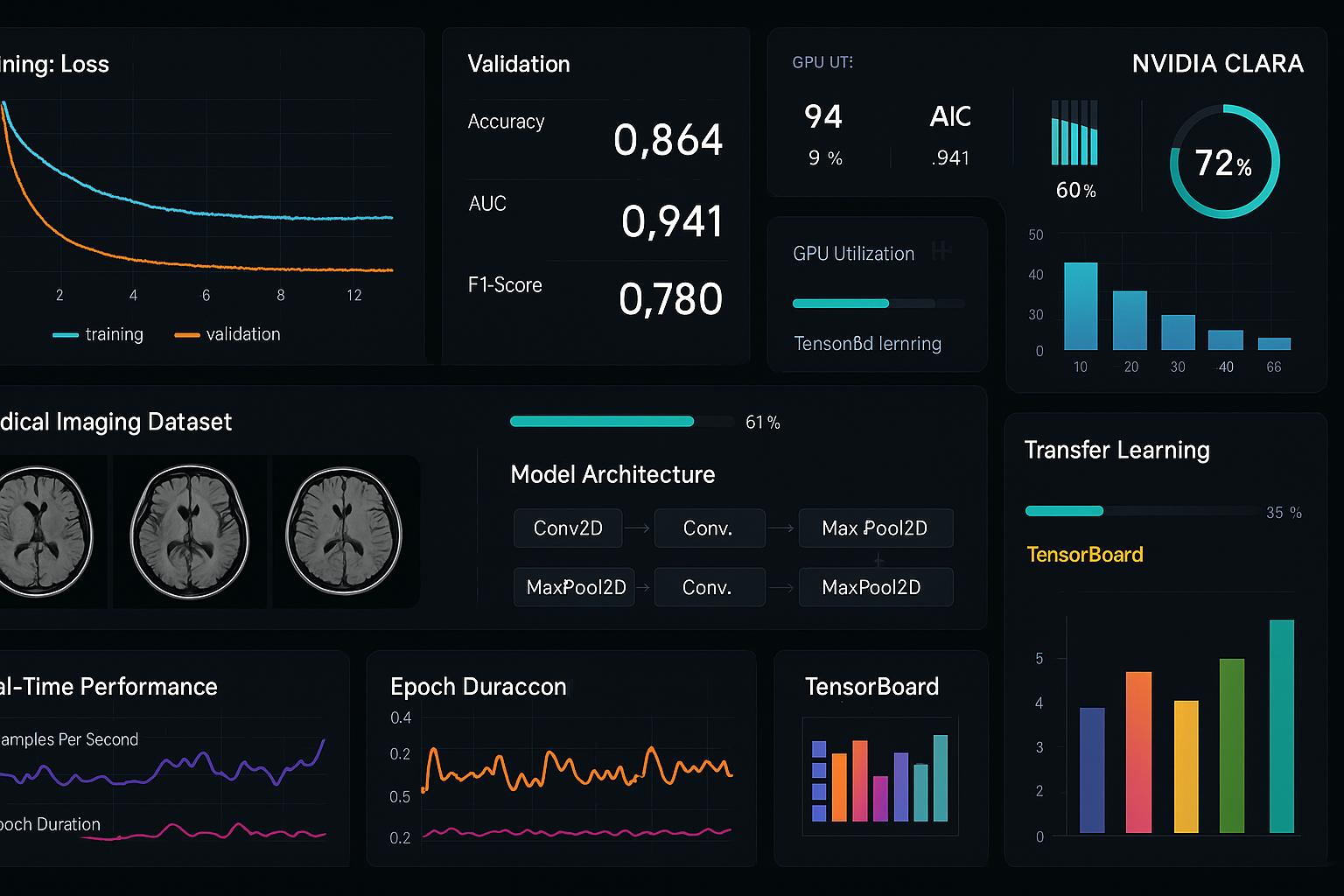
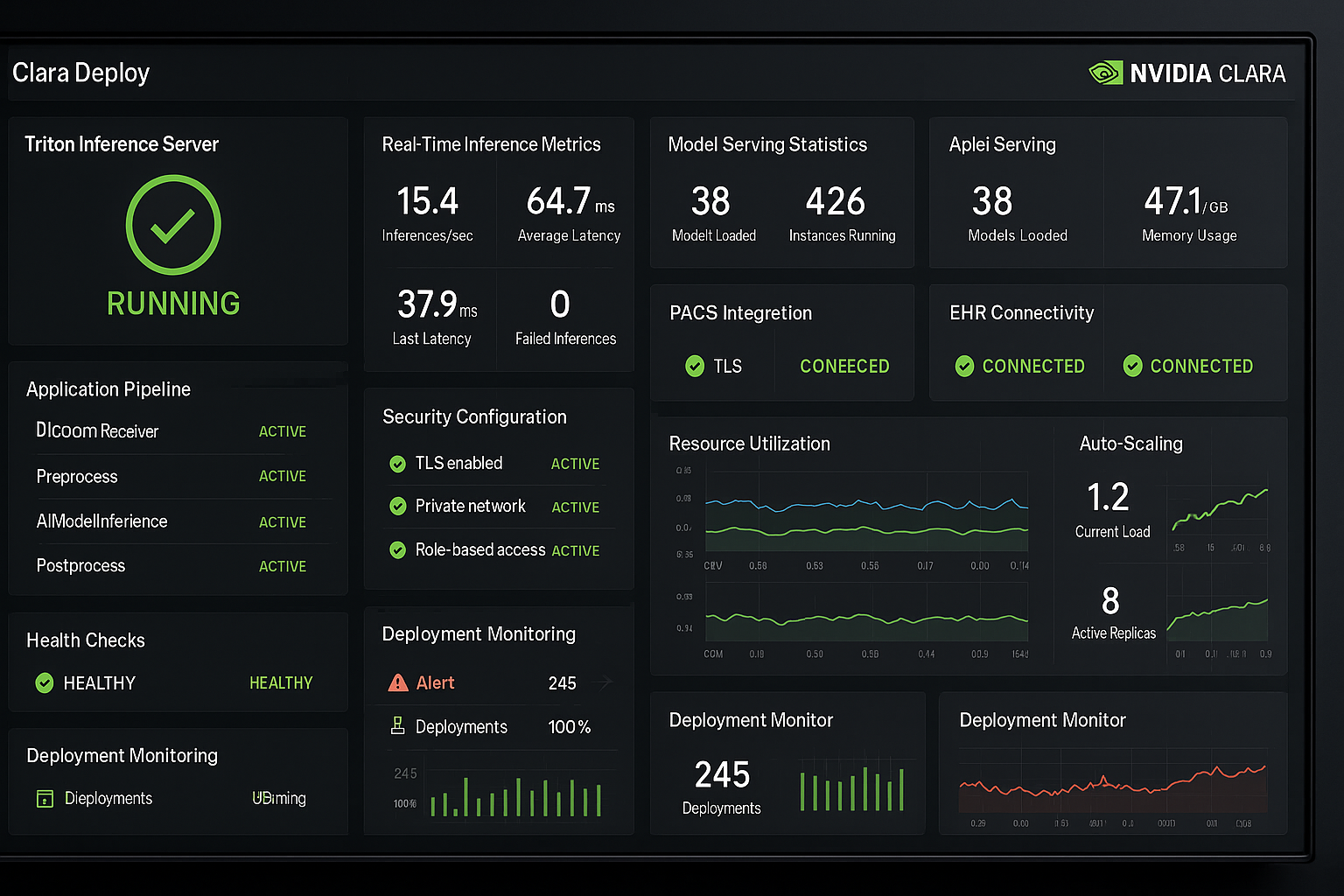



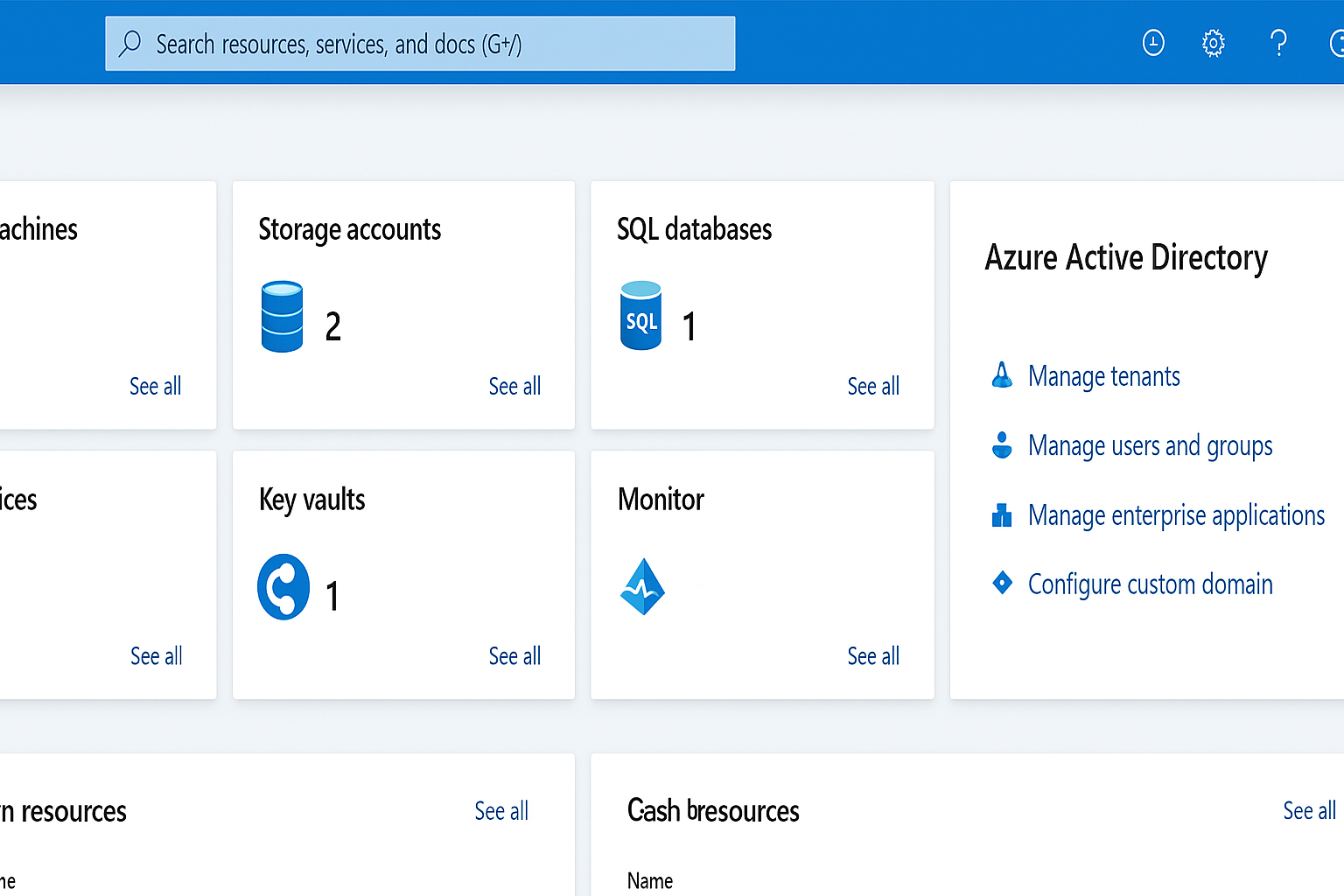 Ultra-wide panoramic view of complete Azure Portal dashboard with all services visible
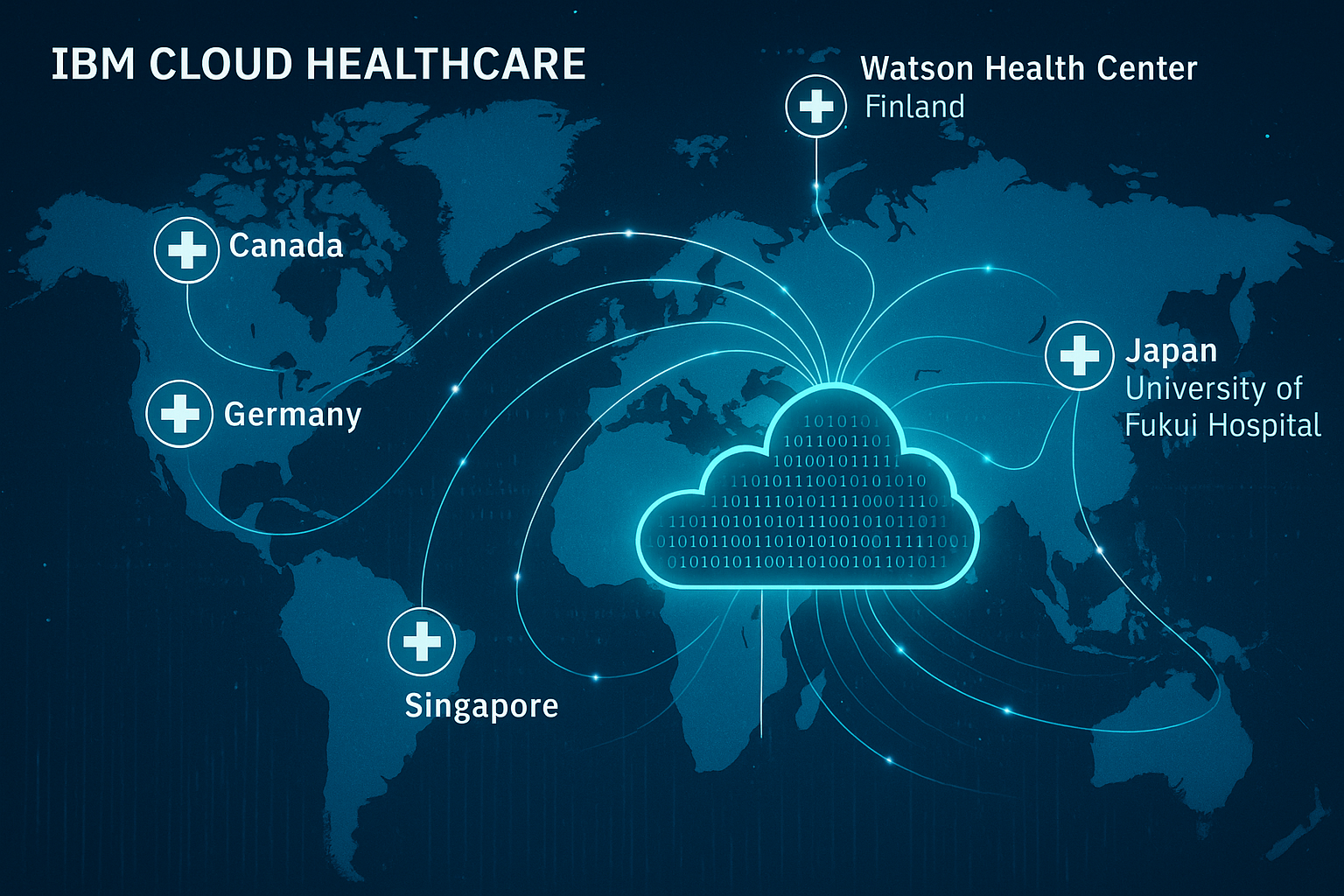
Ultra-wide panoramic view of complete Azure Portal dashboard with all services visible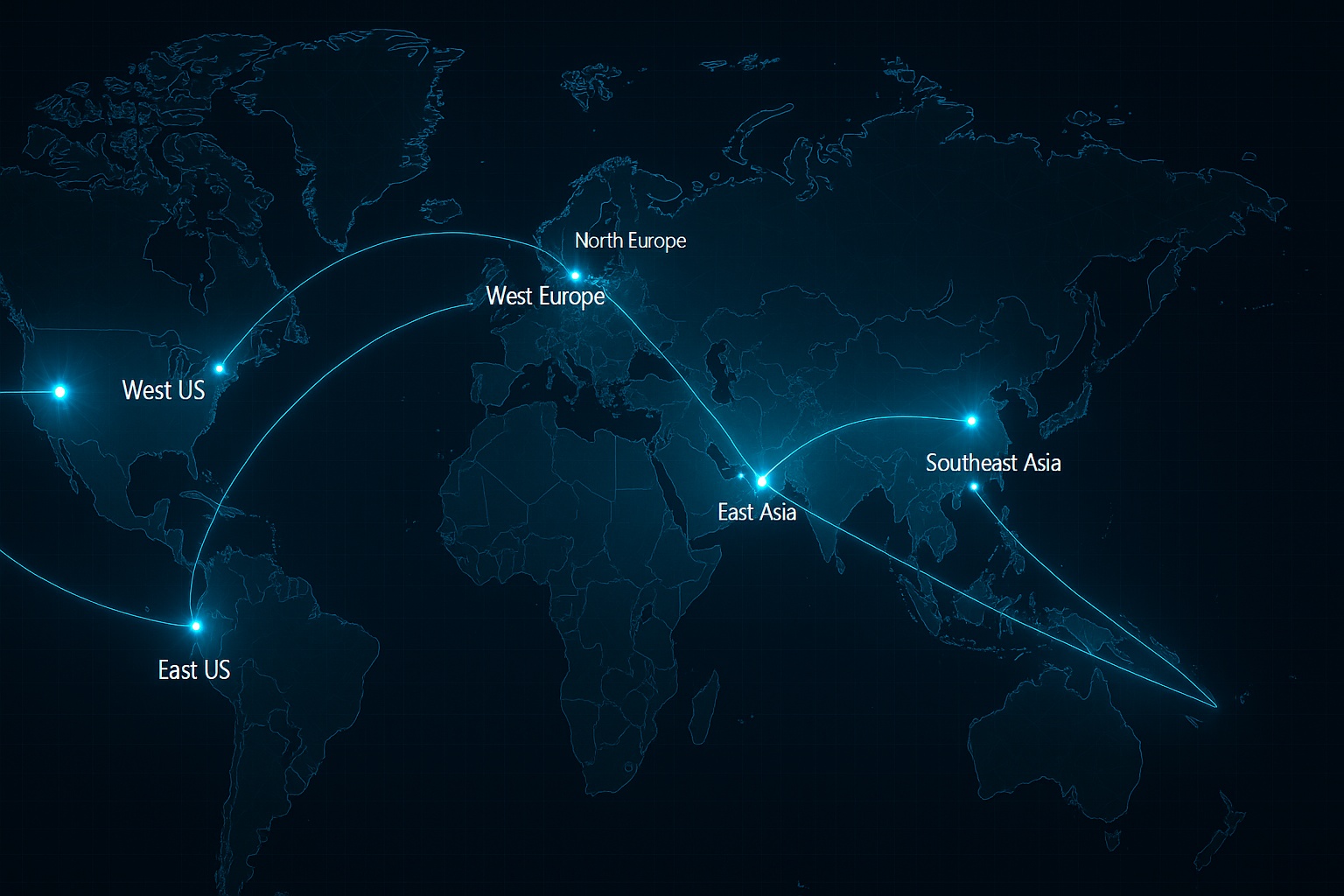 Interactive world map showing all Azure regions with network connections
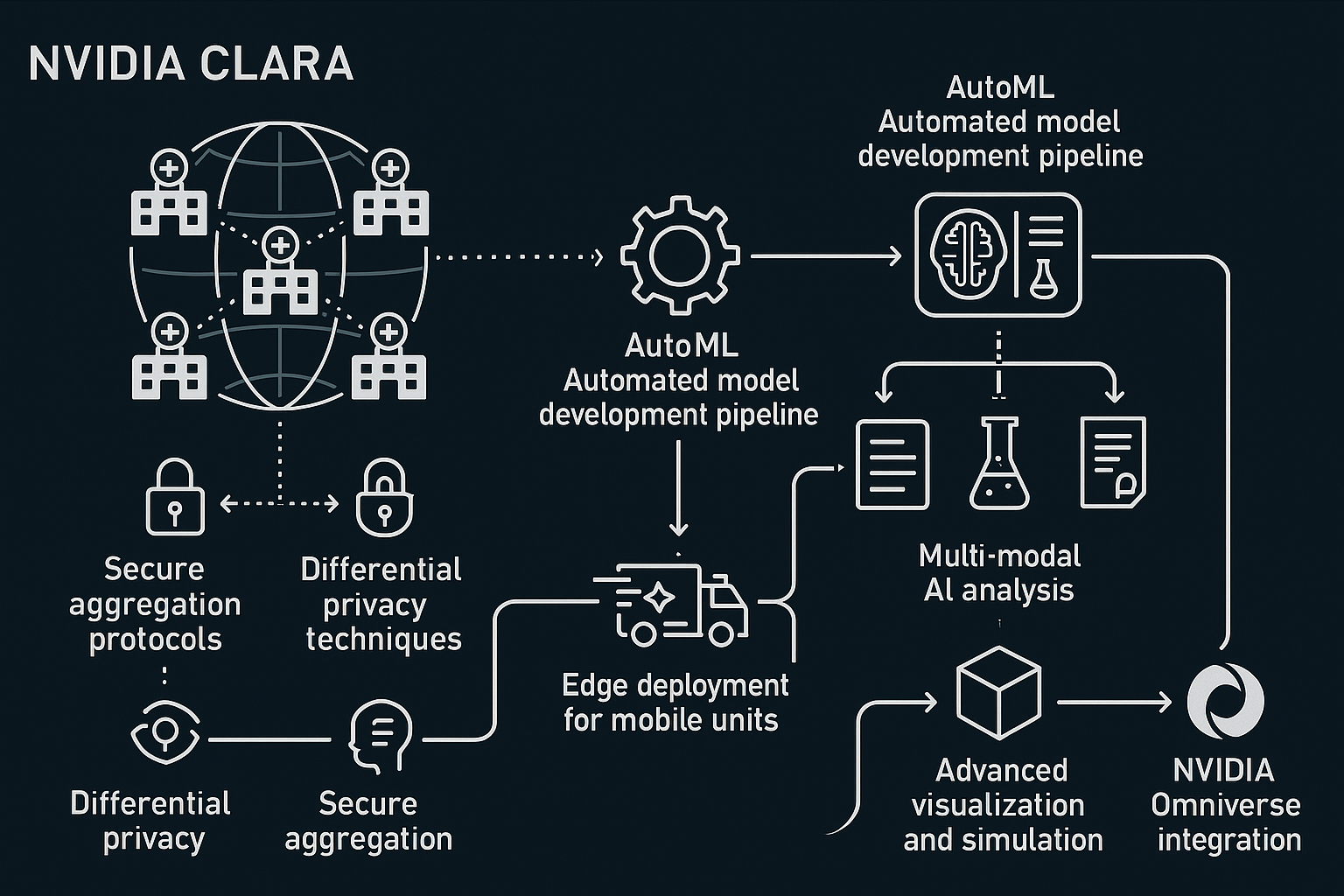
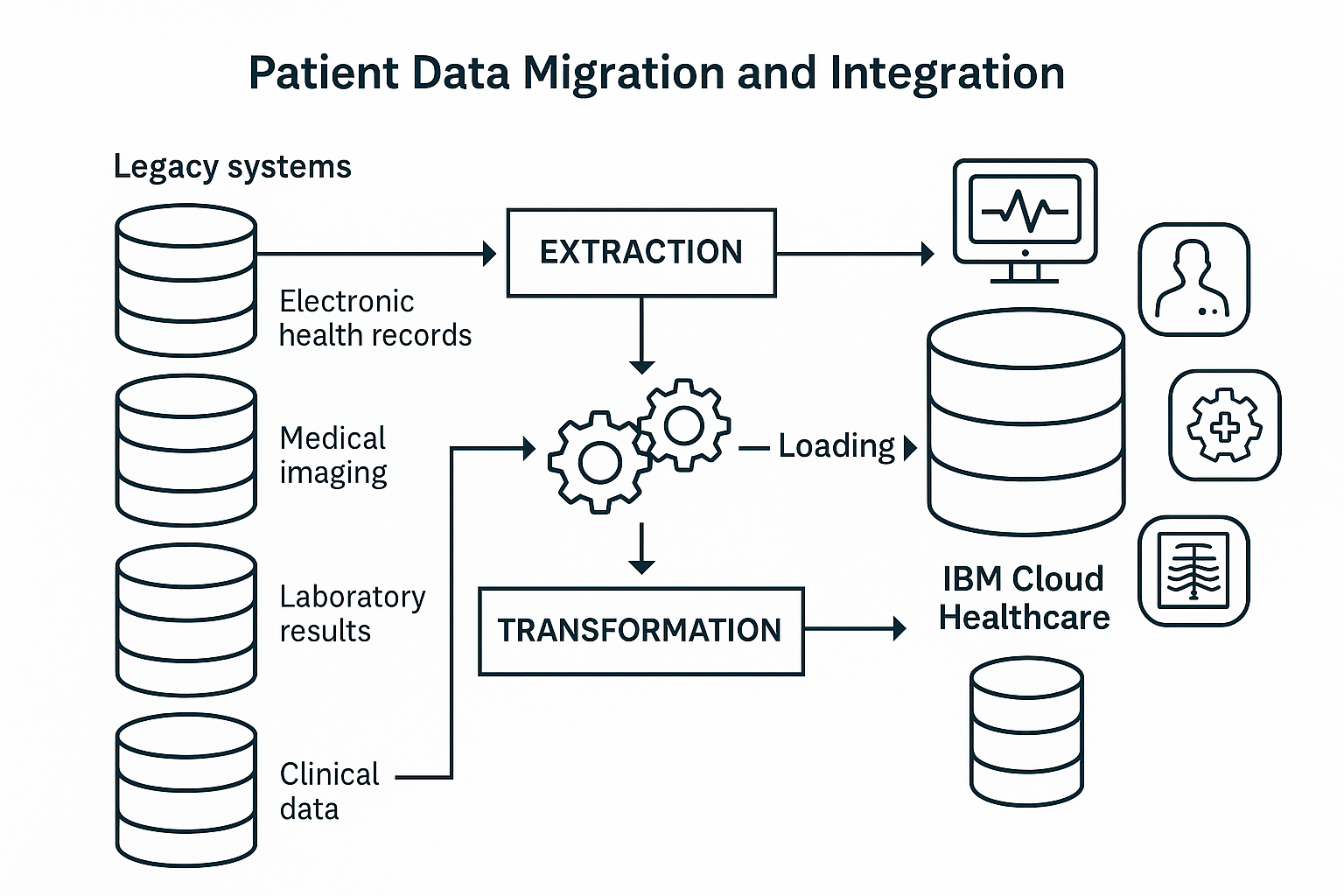
Interactive world map showing all Azure regions with network connections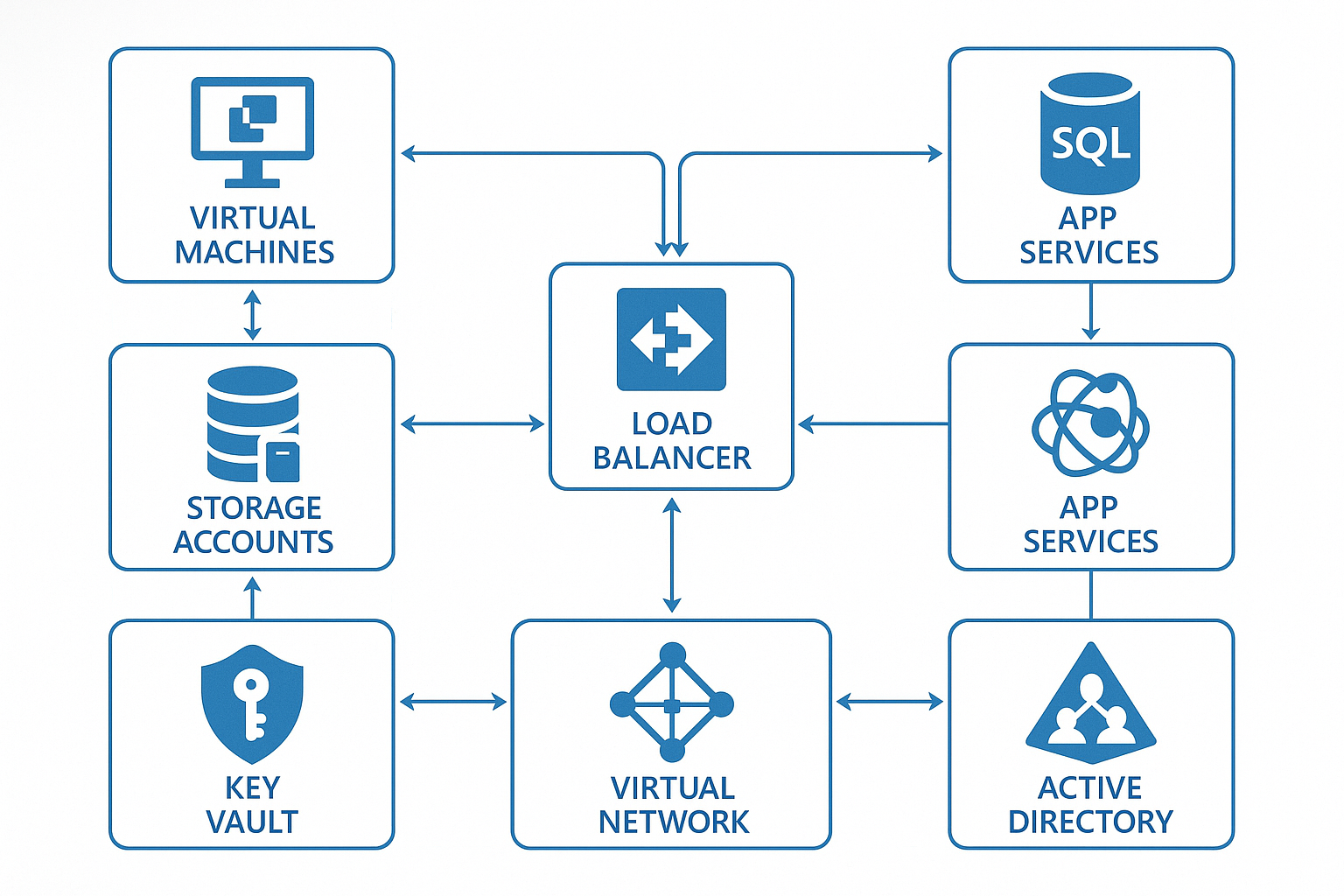 Complete technical diagram showing interconnected Azure services
Complete technical diagram showing interconnected Azure services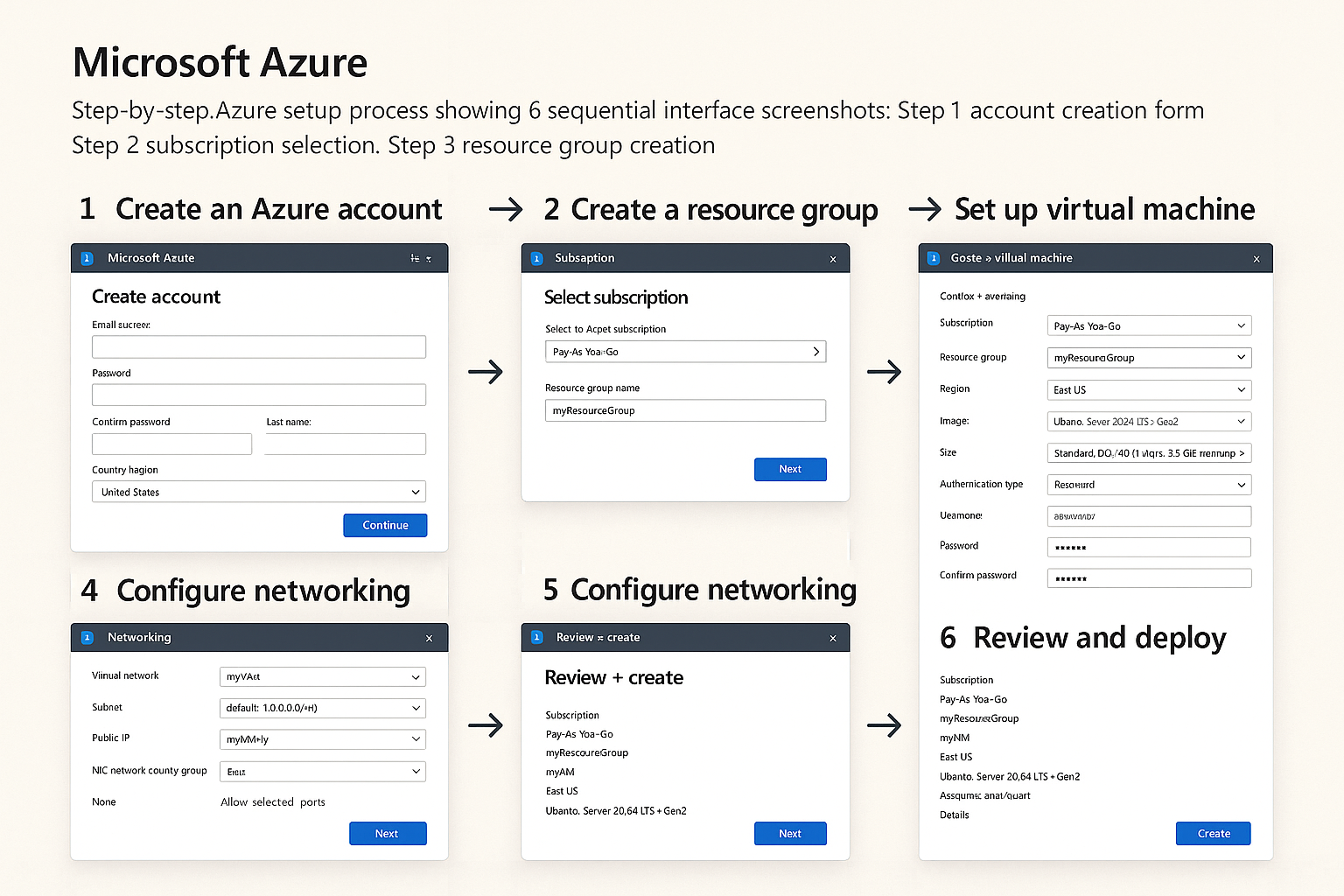 Sequential interface screenshots showing complete Azure setup process
Sequential interface screenshots showing complete Azure setup process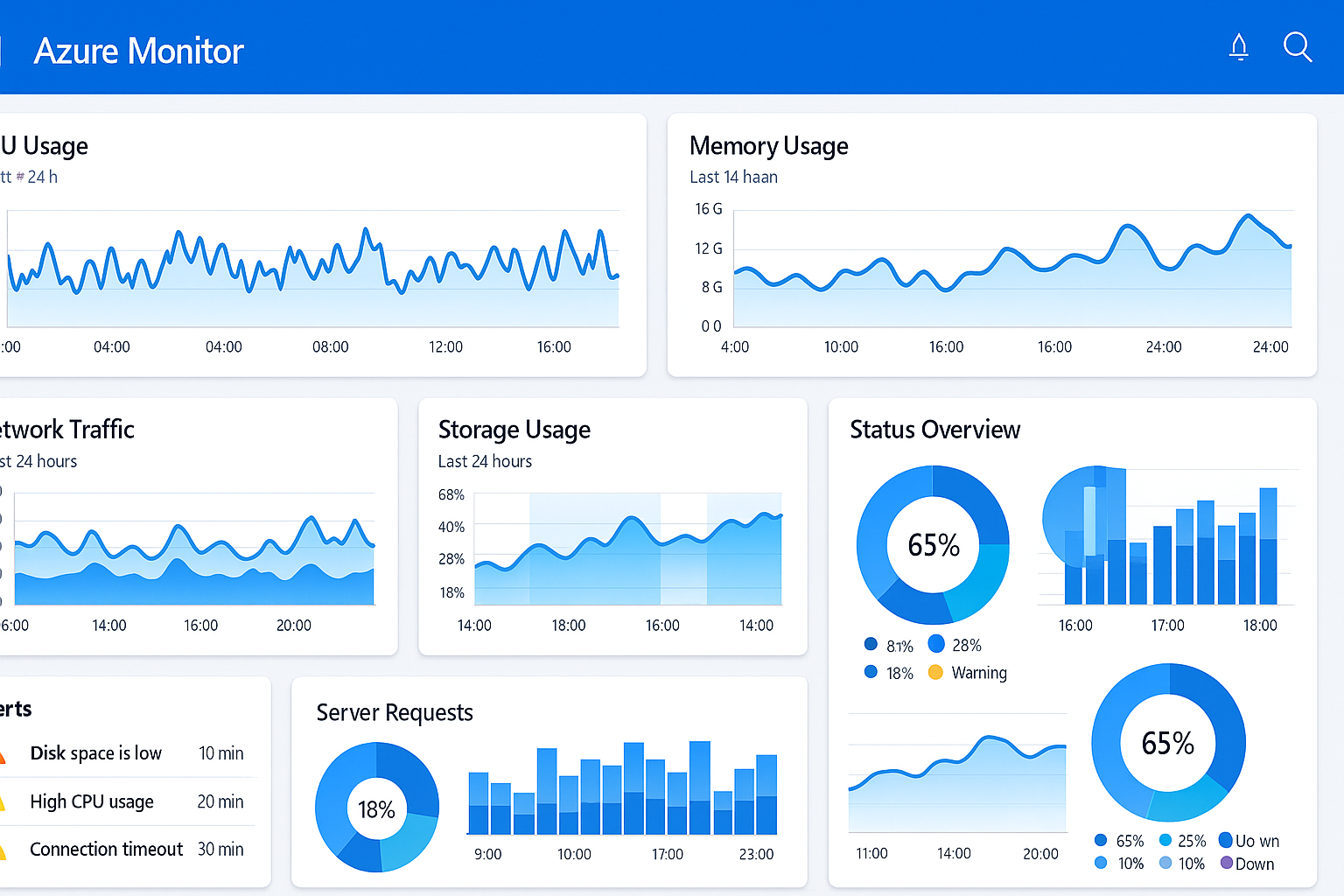 Comprehensive monitoring interface with performance metrics and alerts
Comprehensive monitoring interface with performance metrics and alerts
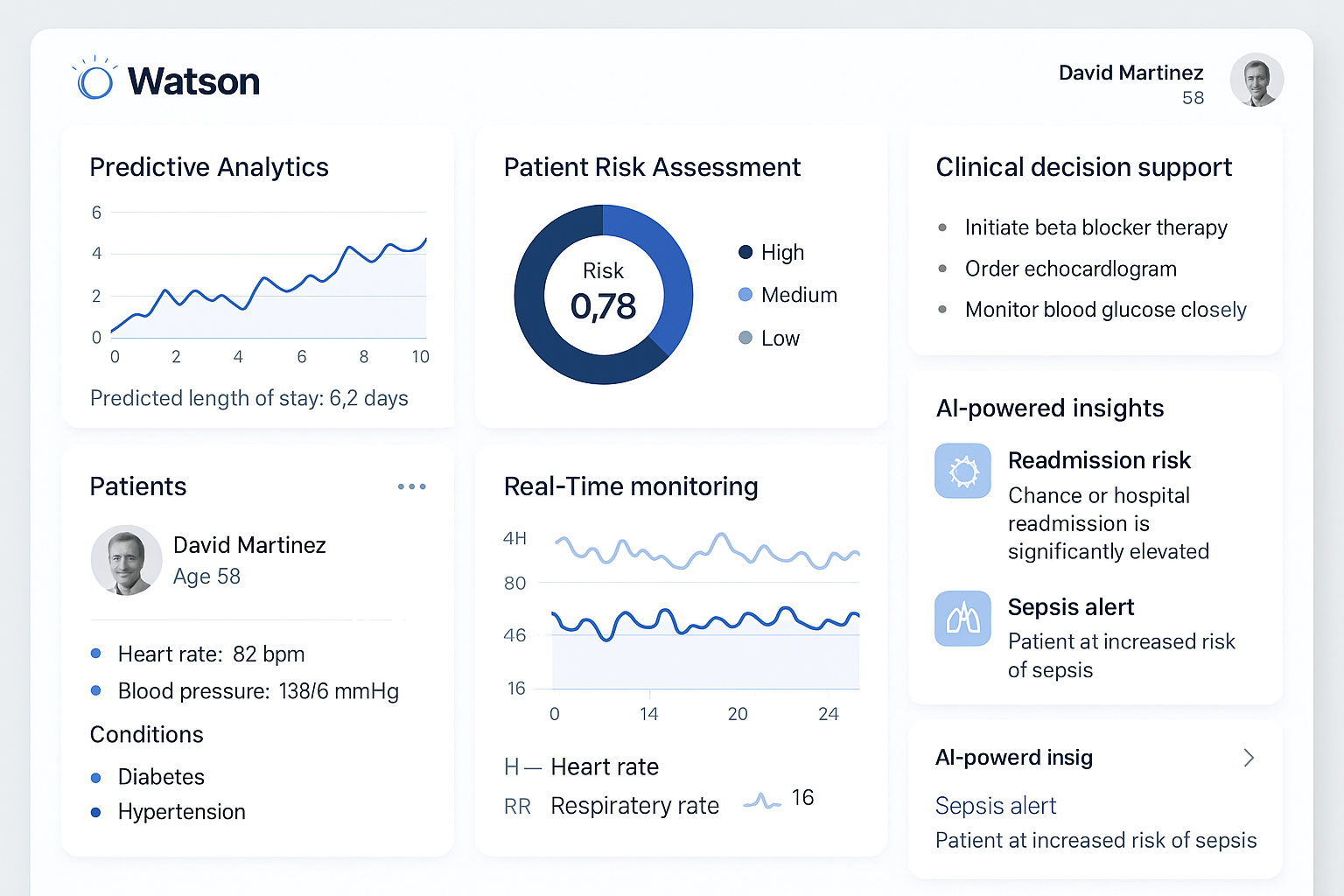
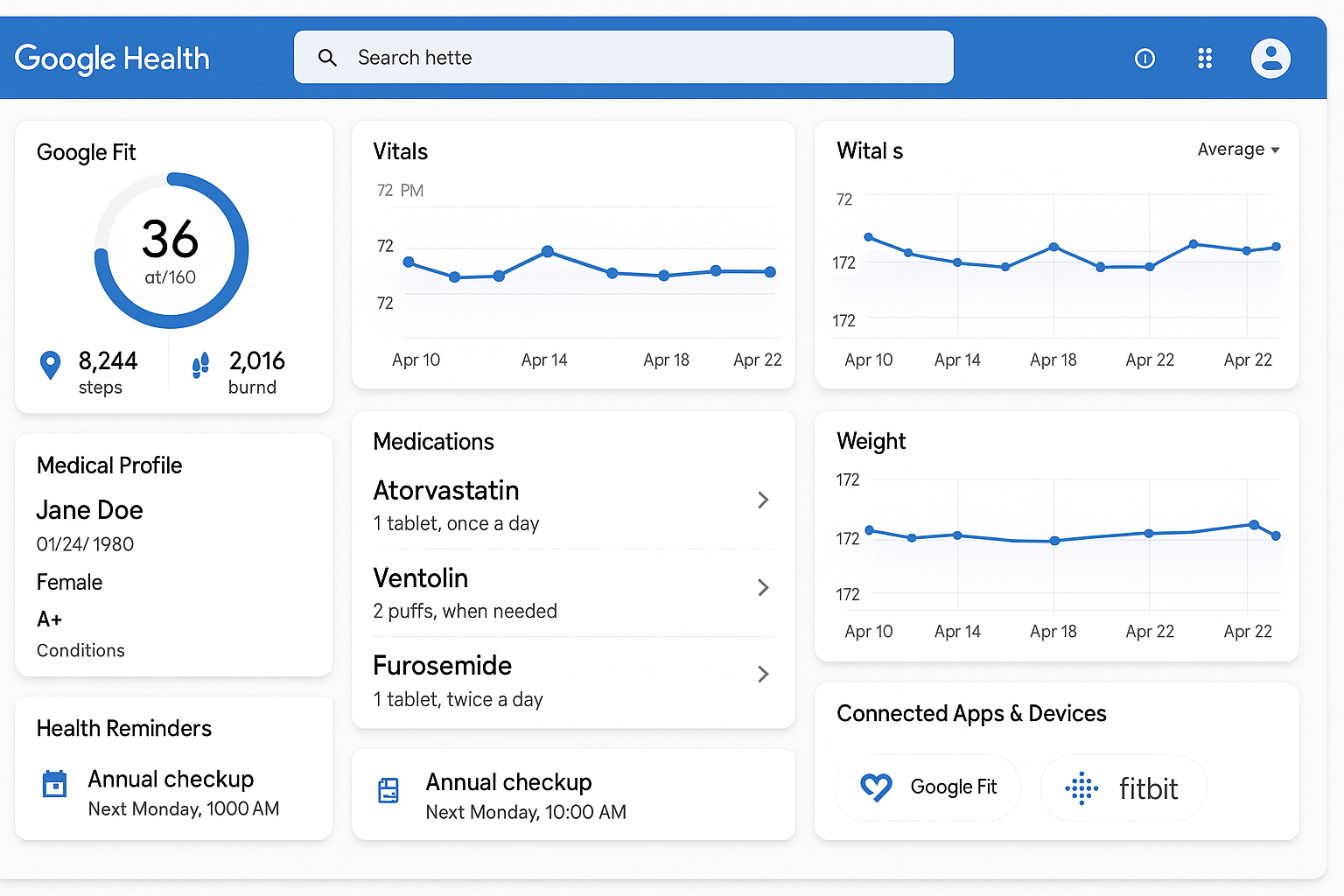 Figure 1: Complete Google Health Dashboard showing all major features including Google Fit integration, health monitoring, and device connectivity
Figure 1: Complete Google Health Dashboard showing all major features including Google Fit integration, health monitoring, and device connectivity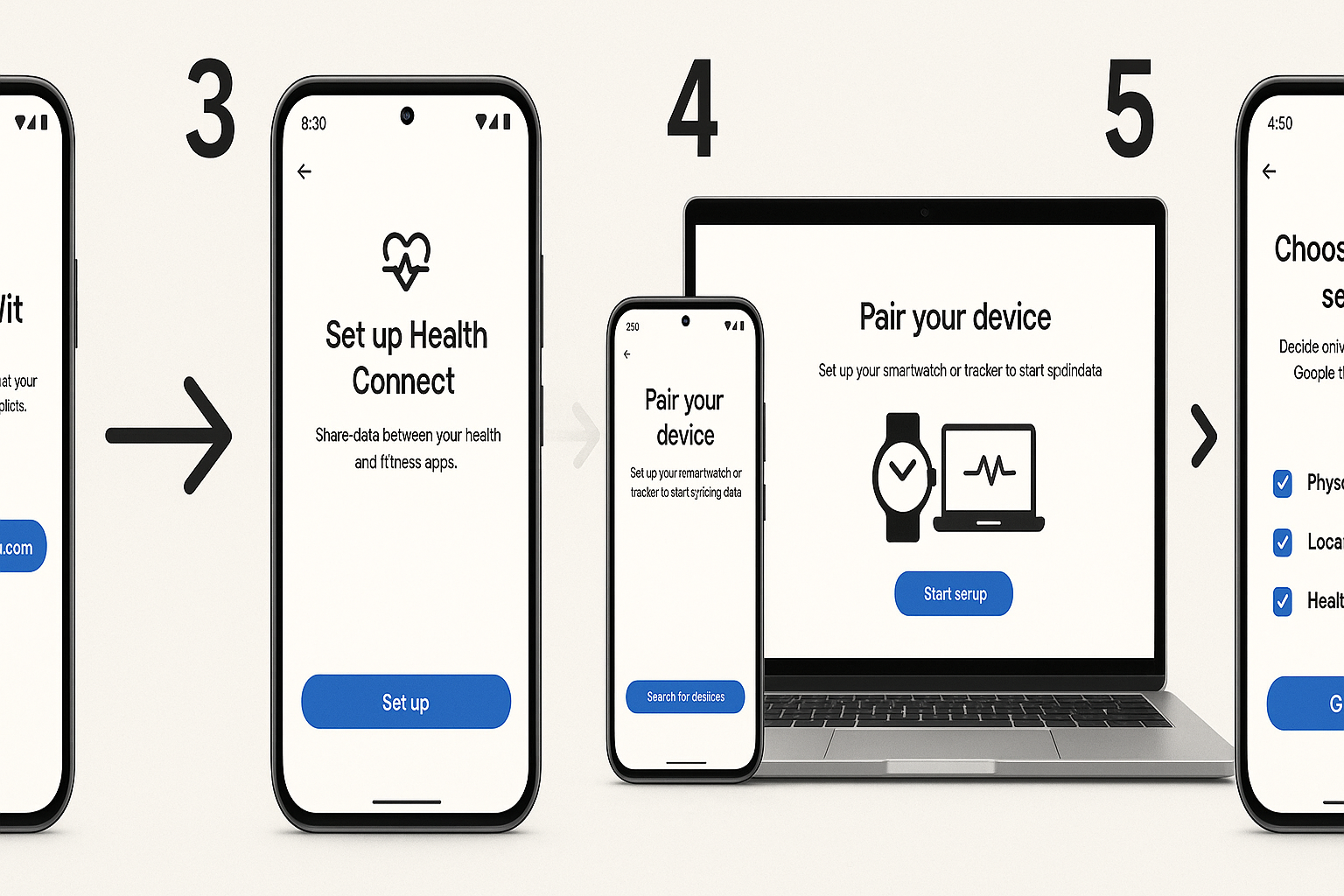 Figure 2: Complete 6-step setup sequence for Google Health: Account configuration, app installation, Health Connect setup, device pairing, privacy settings, and healthcare provider connection
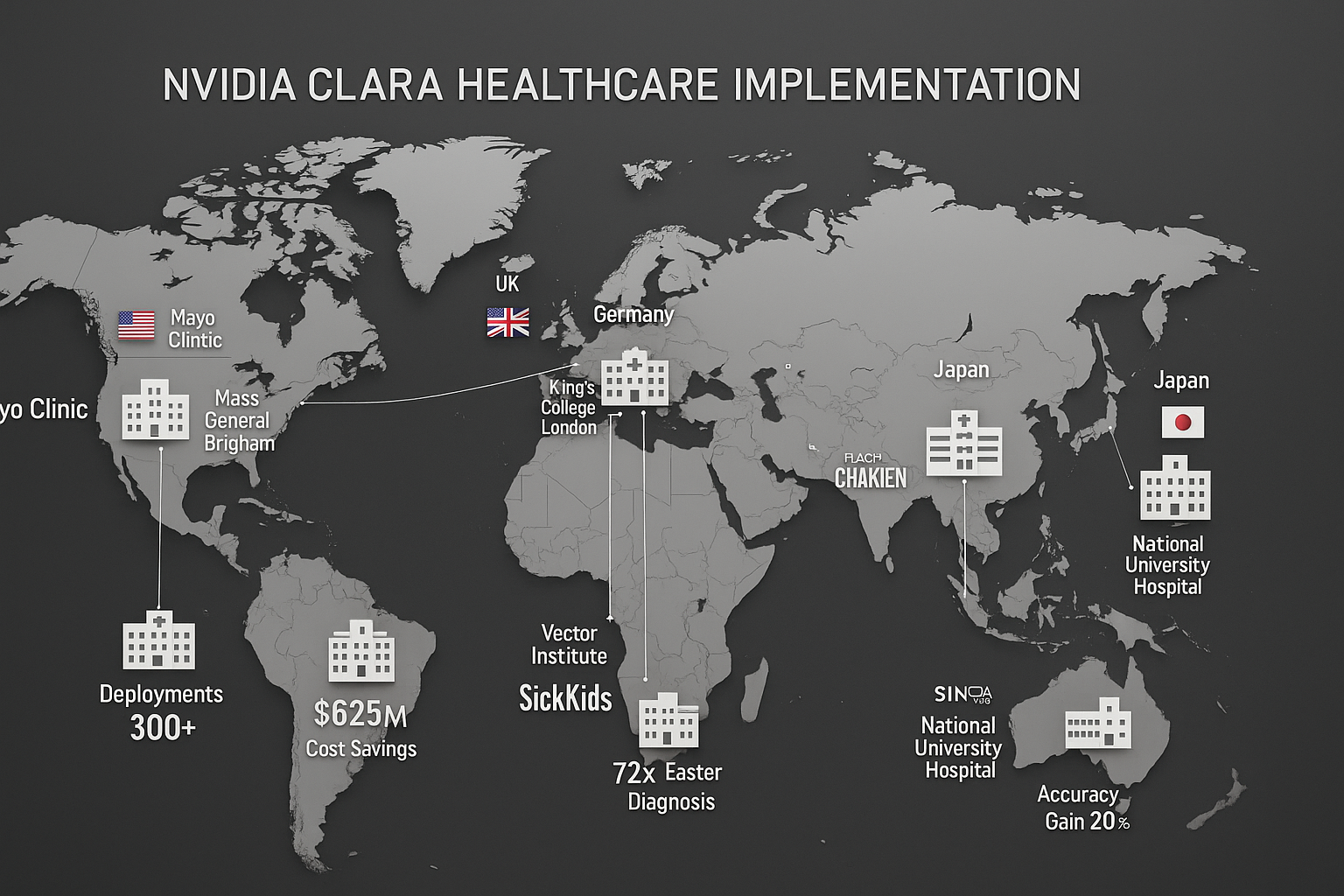
Figure 2: Complete 6-step setup sequence for Google Health: Account configuration, app installation, Health Connect setup, device pairing, privacy settings, and healthcare provider connection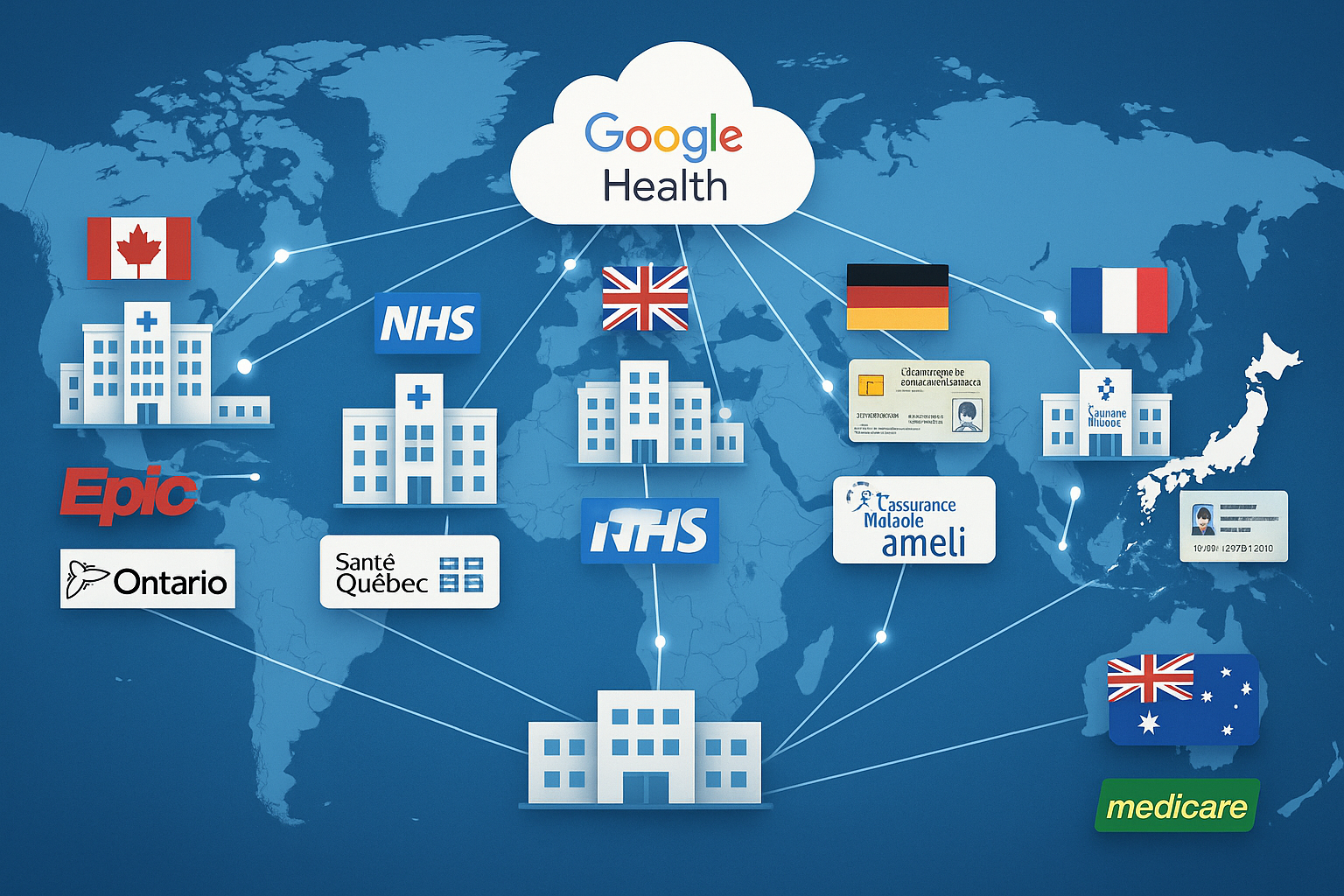 Figure 3: World map showing Google Health integration with healthcare systems across 7 countries: US (Epic/Cerner), UK (NHS), Canada (Provincial Systems), Australia (Medicare), Germany (eGK), Japan (My Number), France (Ameli)
Figure 3: World map showing Google Health integration with healthcare systems across 7 countries: US (Epic/Cerner), UK (NHS), Canada (Provincial Systems), Australia (Medicare), Germany (eGK), Japan (My Number), France (Ameli) Figure 4: Complete connected health ecosystem showing smartphones, smartwatches, fitness trackers, smart scales, blood pressure monitors, and glucose meters all syncing with Google Health cloud platform
Figure 4: Complete connected health ecosystem showing smartphones, smartwatches, fitness trackers, smart scales, blood pressure monitors, and glucose meters all syncing with Google Health cloud platform Figure 5: Advanced security and privacy settings interface showing two-factor authentication, data encryption controls, GDPR compliance settings, and healthcare provider access management
Figure 5: Advanced security and privacy settings interface showing two-factor authentication, data encryption controls, GDPR compliance settings, and healthcare provider access management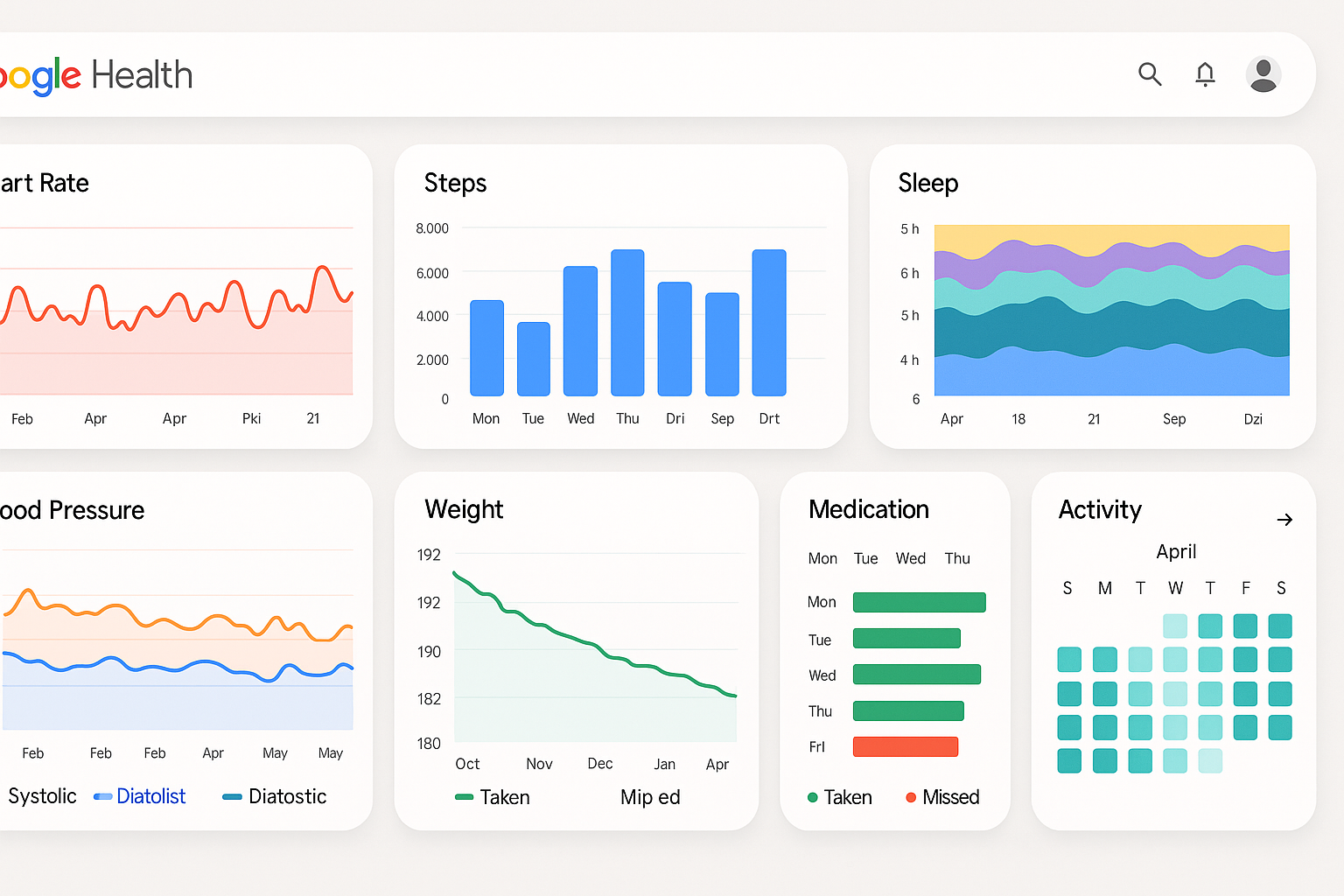 Figure 6: Comprehensive health analytics dashboard displaying heart rate trends, sleep analysis, activity tracking, blood pressure monitoring, and medication adherence with interactive charts and visualizations
Figure 6: Comprehensive health analytics dashboard displaying heart rate trends, sleep analysis, activity tracking, blood pressure monitoring, and medication adherence with interactive charts and visualizations