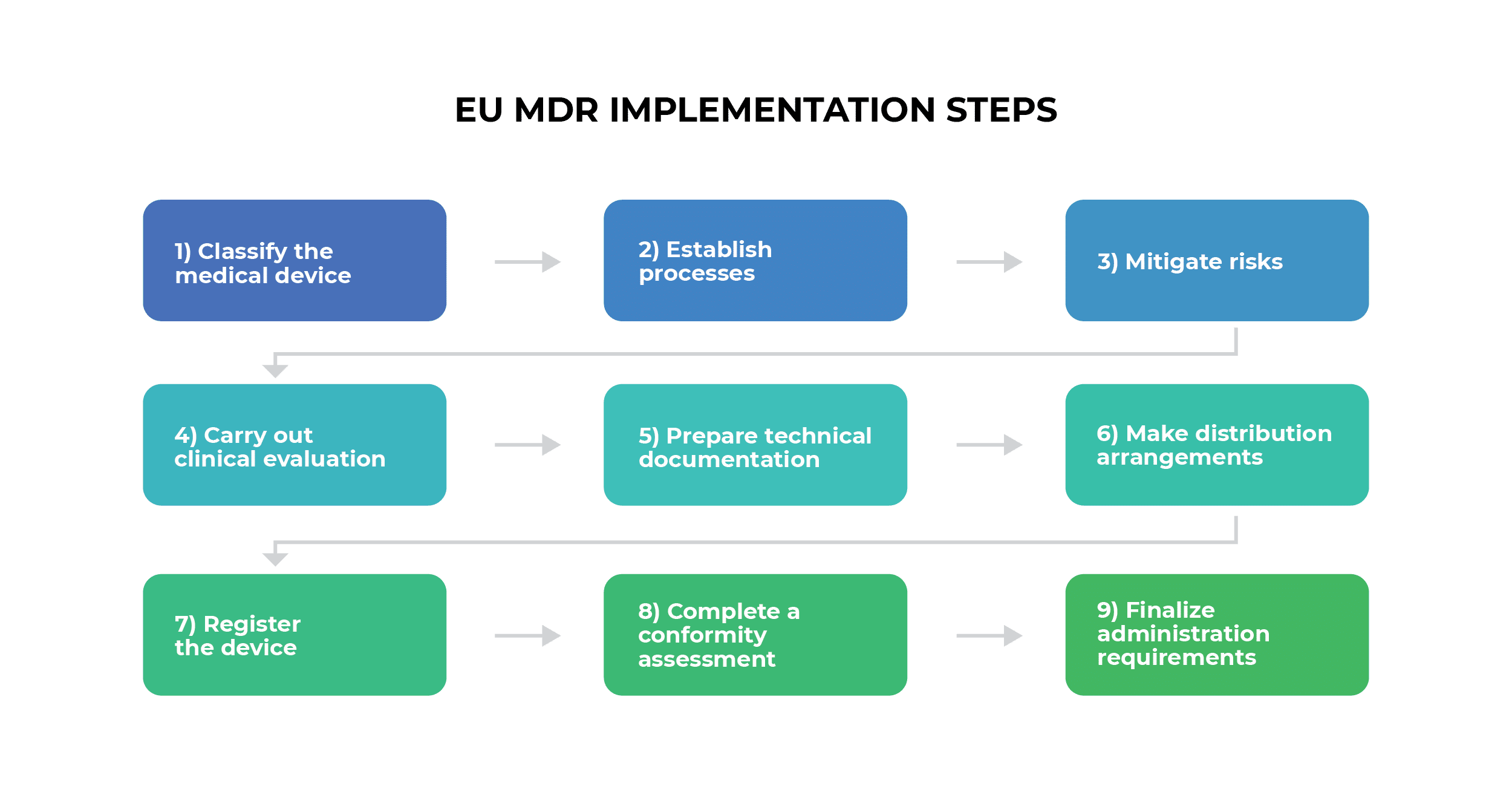
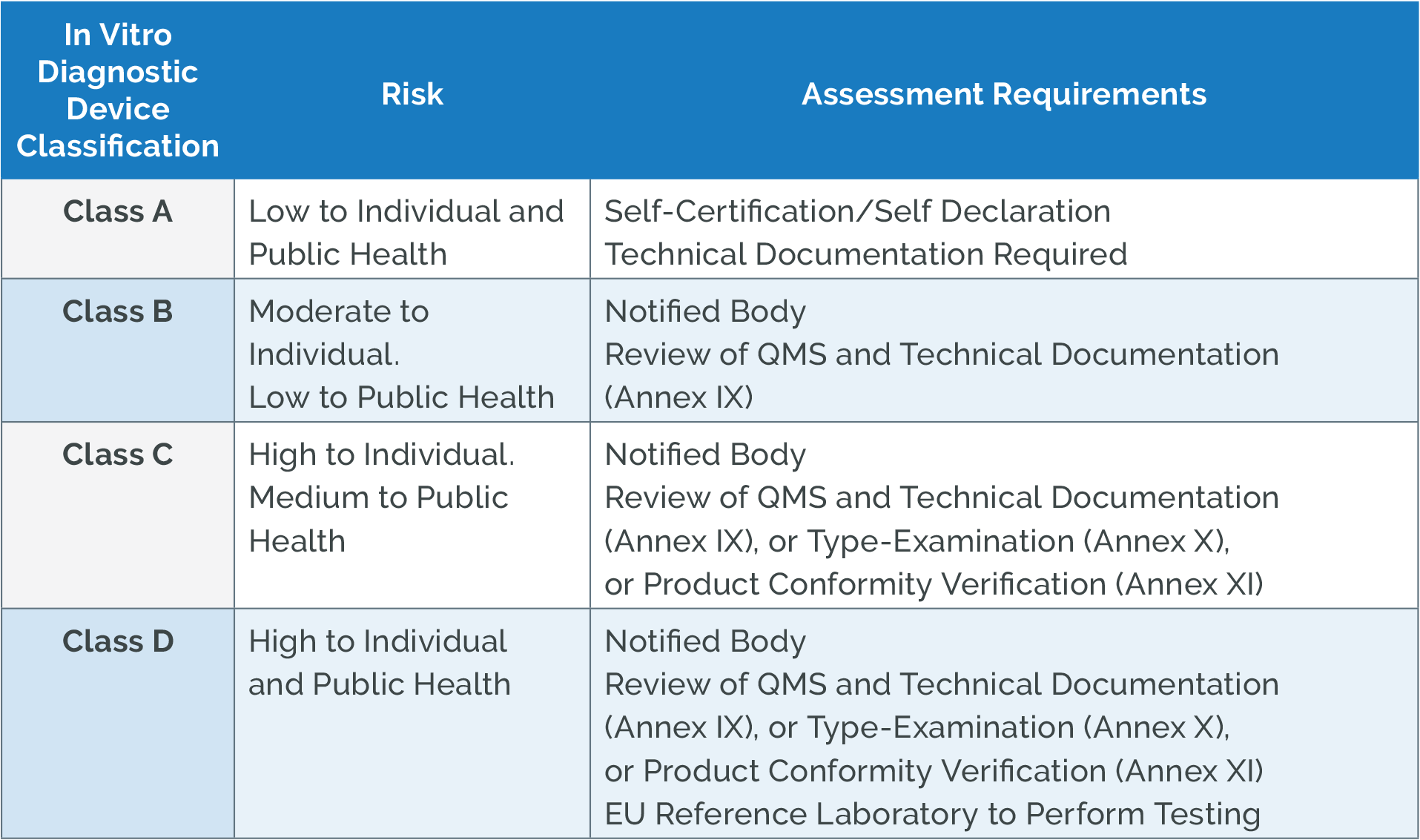
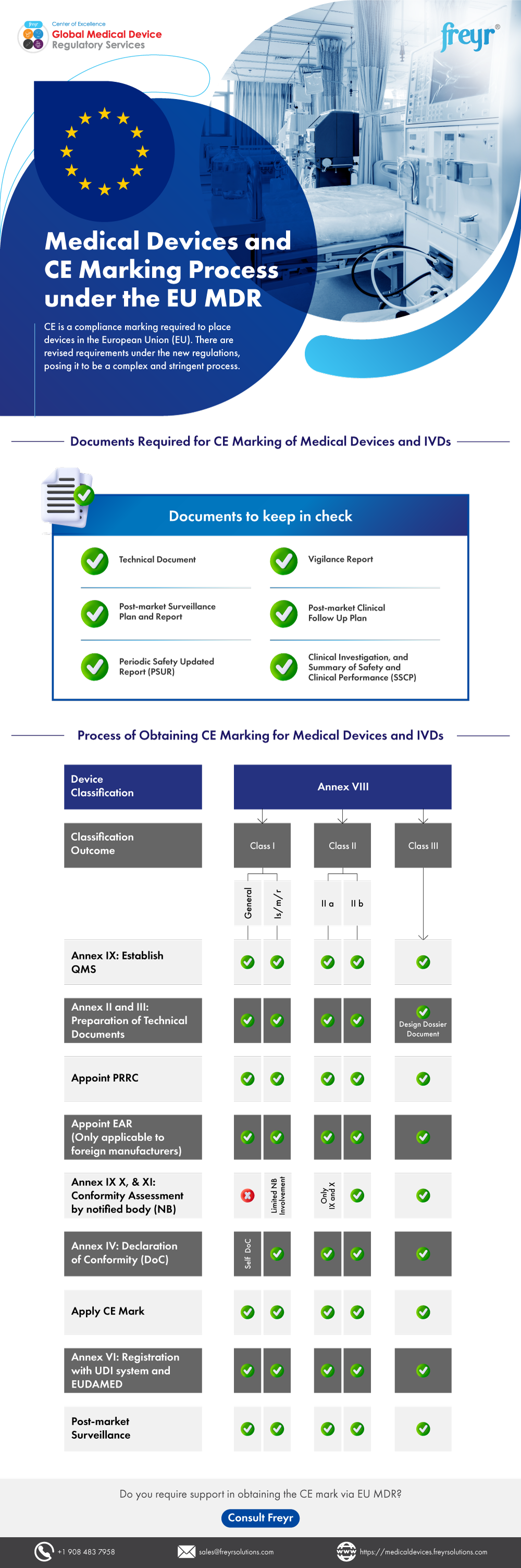
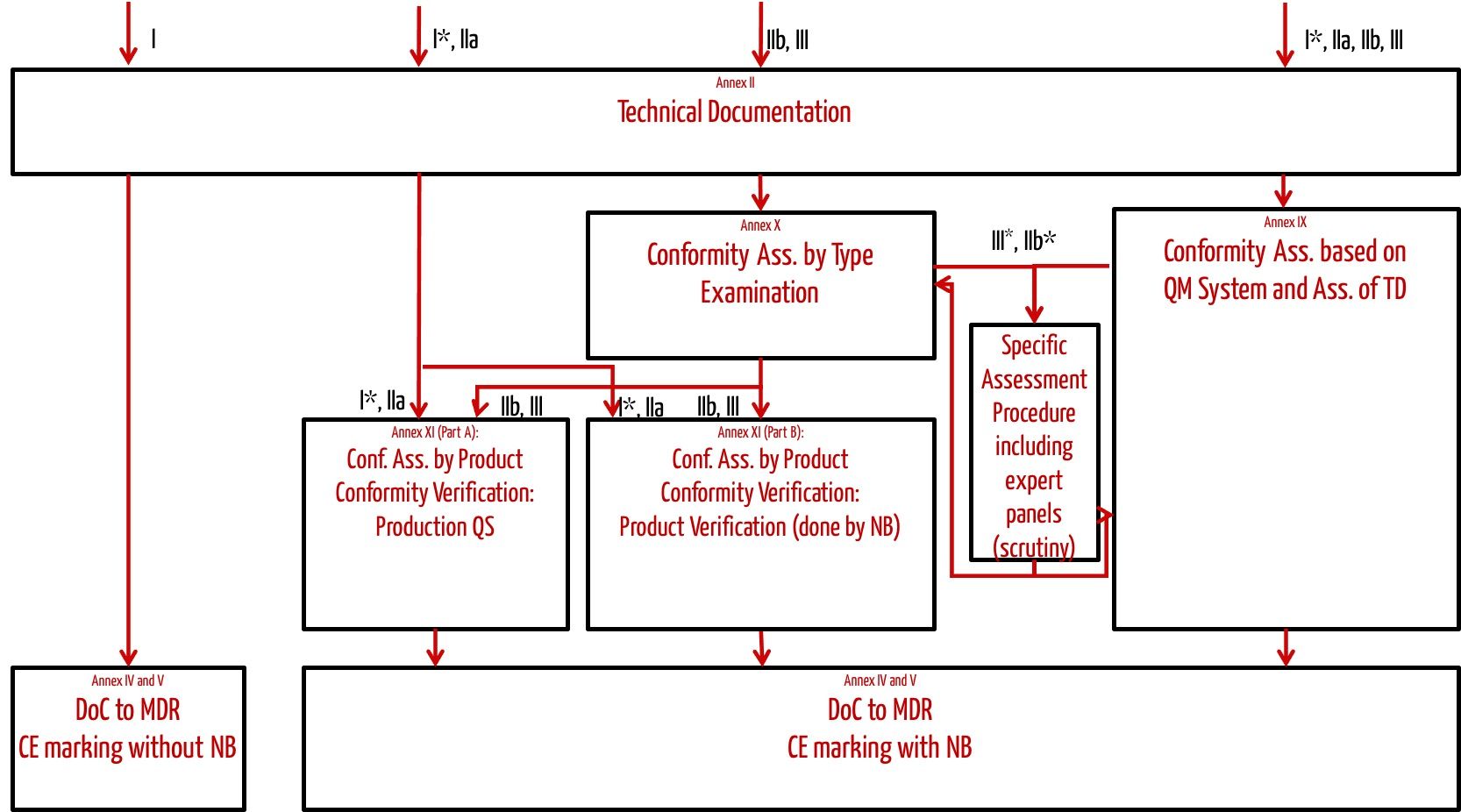
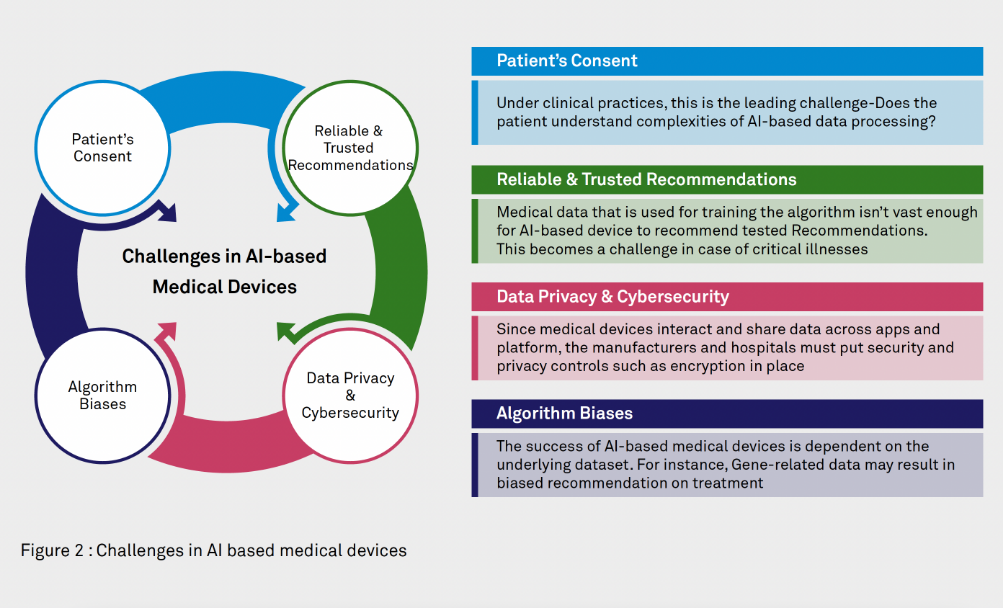
Which Thermometers Are Best for Pediatric Clinics: A Comprehensive Guide to Accurate Temperature Measurement in Children

Temperature measurement is one of the most fundamental vital signs assessed in pediatric healthcare, serving as a critical indicator of a child’s health status and potential presence of infection. For pediatric clinics, selecting the appropriate thermometers is essential for providing accurate, efficient, and child-friendly care. This comprehensive guide examines the various types of thermometers available for pediatric use, their clinical applications, accuracy considerations, and recommendations for different age groups to help healthcare providers make informed decisions about their temperature measurement equipment.
The Critical Importance of Accurate Temperature Measurement in Pediatric Care
Temperature measurement in pediatric patients presents unique challenges that require specialized consideration. According to Cleveland Clinic research, the accuracy of temperature measurement is largely affected by a child’s ability to cooperate with the measurement process, making age-appropriate thermometer selection crucial for reliable results.
Age-Specific Considerations
Different age groups require different approaches to temperature measurement:
- Infants (0-6 months): Require non-invasive or minimally invasive methods due to inability to cooperate
- Toddlers (6 months-3 years): Need quick, comfortable measurement methods to minimize distress
- School-age children (4+ years): Can cooperate with more traditional measurement methods
- Adolescents: Can use adult-appropriate thermometers with proper instruction
Clinical Significance
Accurate temperature measurement is particularly critical in pediatric settings for several reasons:
- Fever detection in immunocompromised children can be life-saving
- Hypothermia identification in newborns requires precise measurement
- Medication dosing often depends on accurate temperature readings
- Treatment decisions are frequently based on fever presence and severity
Digital Thermometers: The Foundation of Pediatric Temperature Measurement
Digital thermometers represent the most versatile and widely used category of temperature measurement devices in pediatric clinics. These electronic devices use thermistors or other temperature-sensitive sensors to provide quick, accurate readings.
Standard Digital Thermometers
Standard digital thermometers feature a small metal tip that can be used for multiple measurement sites, making them highly versatile for pediatric applications.
Clinical Applications:
- Oral temperature measurement for children aged 4 and older
- Rectal temperature measurement for infants and young children (most accurate method)
- Axillary (armpit) measurement for screening purposes across all age groups
Advantages:
- Cost-effective and reliable
- Quick measurement time (10-60 seconds)
- Easy to clean and maintain
- Widely available and FDA-approved
Limitations:
- Requires patient cooperation for oral use
- May cause discomfort with rectal measurement
- Less accurate with axillary measurement
Flexible-Tip Digital Thermometers

Flexible-tip digital thermometers are specifically designed for pediatric use, featuring soft, bendable tips that provide enhanced comfort and safety for young patients.
Key Features:
- Soft, flexible probe tip reduces discomfort during rectal measurement
- Quick measurement time (8-10 seconds typical)
- Large, easy-to-read displays for healthcare providers
- Fever alert indicators with color-coded or audible warnings
Clinical Benefits:
- Reduced patient anxiety and discomfort
- Improved safety for rectal temperature measurement
- Enhanced cooperation from pediatric patients
- Reliable accuracy across multiple measurement sites
Infrared Thermometers: Non-Contact Temperature Measurement

Infrared thermometers have revolutionized pediatric temperature measurement by providing fast, non-contact options that are particularly valuable in clinic settings where speed and infection control are priorities.
Forehead (Temporal Artery) Thermometers
Temporal artery thermometers use infrared technology to measure heat emitted from the temporal artery, which runs across the forehead just beneath the skin surface.

Clinical Advantages:
- Non-invasive measurement eliminates patient discomfort
- Rapid results (1-3 seconds typical measurement time)
- No risk of cross-contamination between patients
- Suitable for all age groups including newborns
Best Practices for Use:
- Ensure forehead is clean and dry before measurement
- Remove hats or headbands that might affect accuracy
- Allow device to acclimate to room temperature
- Take measurements away from direct sunlight or heat sources
Professional-Grade Infrared Thermometers

Professional-grade infrared thermometers designed for clinical use offer enhanced accuracy and durability compared to consumer models.
Advanced Features:
- Medical-grade sensors for enhanced accuracy
- Memory storage for tracking temperature trends
- Adjustable distance measurement for optimal positioning
- Calibration capabilities for maintaining accuracy over time
Clinical Applications:
- High-volume pediatric clinics requiring rapid patient throughput
- Emergency departments with time-critical assessments
- Infection control environments requiring minimal contact
Ear (Tympanic) Thermometers: Proven Accuracy for Pediatric Use

Ear thermometers use infrared technology to measure the temperature of the tympanic membrane, which receives blood supply from the same arteries that supply the hypothalamus, providing an accurate reflection of core body temperature.
Clinical Evidence and Accuracy
Research published in PMC medical journals indicates that tympanic thermometers provide accurate core temperature measurements that correlate closely with pulmonary artery temperatures, even during rapid temperature changes.
Age-Specific Recommendations:
- 6 months and older: Optimal accuracy and ease of use
- Under 6 months: May be less reliable due to small ear canal size
- Adults and older children: Excellent accuracy and patient acceptance
Professional Tympanic Thermometers

Professional-grade tympanic thermometers offer enhanced features specifically designed for clinical environments.
Advanced Capabilities:
- Pre-warmed tips for patient comfort and accuracy
- ExacTemp technology ensures proper positioning
- Professional accuracy within ±0.2°C of core temperature
- Disposable probe covers for infection control
Clinical Benefits:
- Fast measurement (1-2 seconds)
- High patient acceptance across age groups
- Reliable accuracy when properly positioned
- Reduced risk of cross-contamination
Temporal Artery Thermometers: The Gold Standard for Non-Invasive Measurement

Temporal artery thermometers represent advanced infrared technology specifically designed to measure the temperature of the temporal artery, providing highly accurate core temperature readings.
Professional Temporal Artery Systems

Professional temporal artery thermometers like the Exergen TemporalScanner series are widely used in top pediatric hospitals across the United States.
Clinical Advantages:
- Arterial heat balance technology for enhanced accuracy
- Gentle forehead scanning motion for patient comfort
- Age-neutral performance from newborns to adults
- Clinical validation through extensive peer-reviewed research
Operational Benefits:
- Reduces nursing time compared to traditional methods
- Eliminates need for probe covers
- Provides consistent accuracy across operators
- Suitable for high-volume clinical environments
Specialized Pediatric Thermometer Features
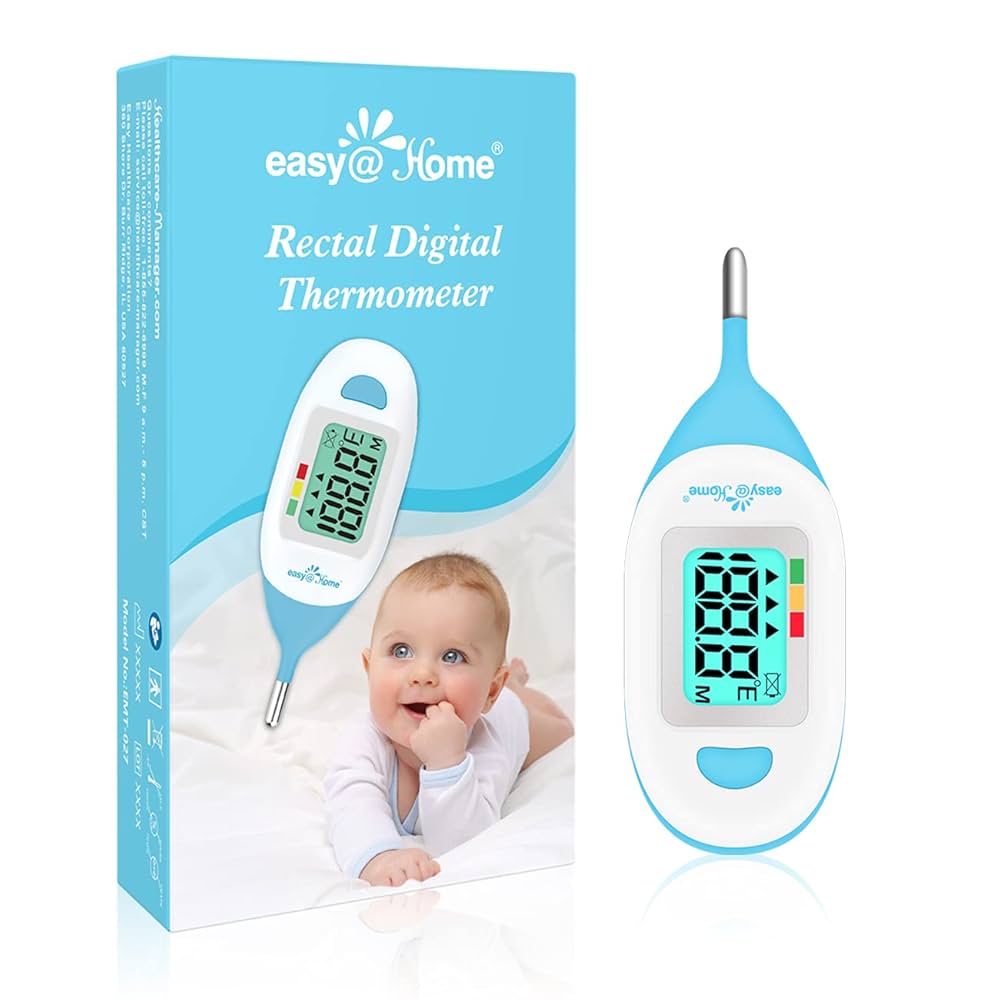
Modern pediatric thermometers incorporate specialized features designed to address the unique challenges of measuring temperature in children.
Age-Appropriate Design Elements
Infant-Specific Features:
- Short, stubby probes designed for small anatomy
- Flexible tips for safety and comfort
- Quick measurement times to minimize distress
- Large display screens for easy reading in low light
Toddler-Friendly Features:
- Colorful, engaging designs to reduce anxiety
- Fun sounds or lights to distract during measurement
- Durable construction to withstand drops and impacts
- Easy-grip handles for secure operation
Smart Technology Integration
:max_bytes(150000):strip_icc()/prt-munchkin-mini-thermometer-21-55beb9483ab84a5da7caafd27163f854.jpeg)
Modern pediatric thermometers increasingly incorporate smart technology features:
Connected Health Features:
- Smartphone app integration for temperature tracking
- Cloud-based data storage for long-term monitoring
- Alert systems for fever notifications
- Multi-user profiles for family or clinic use
Clinical Documentation:
- Electronic health record integration for seamless documentation
- Automatic time and date stamping for accurate records
- Trend analysis capabilities for pattern recognition
- Export functions for sharing with healthcare providers
Comparative Analysis: Accuracy by Age Group and Method
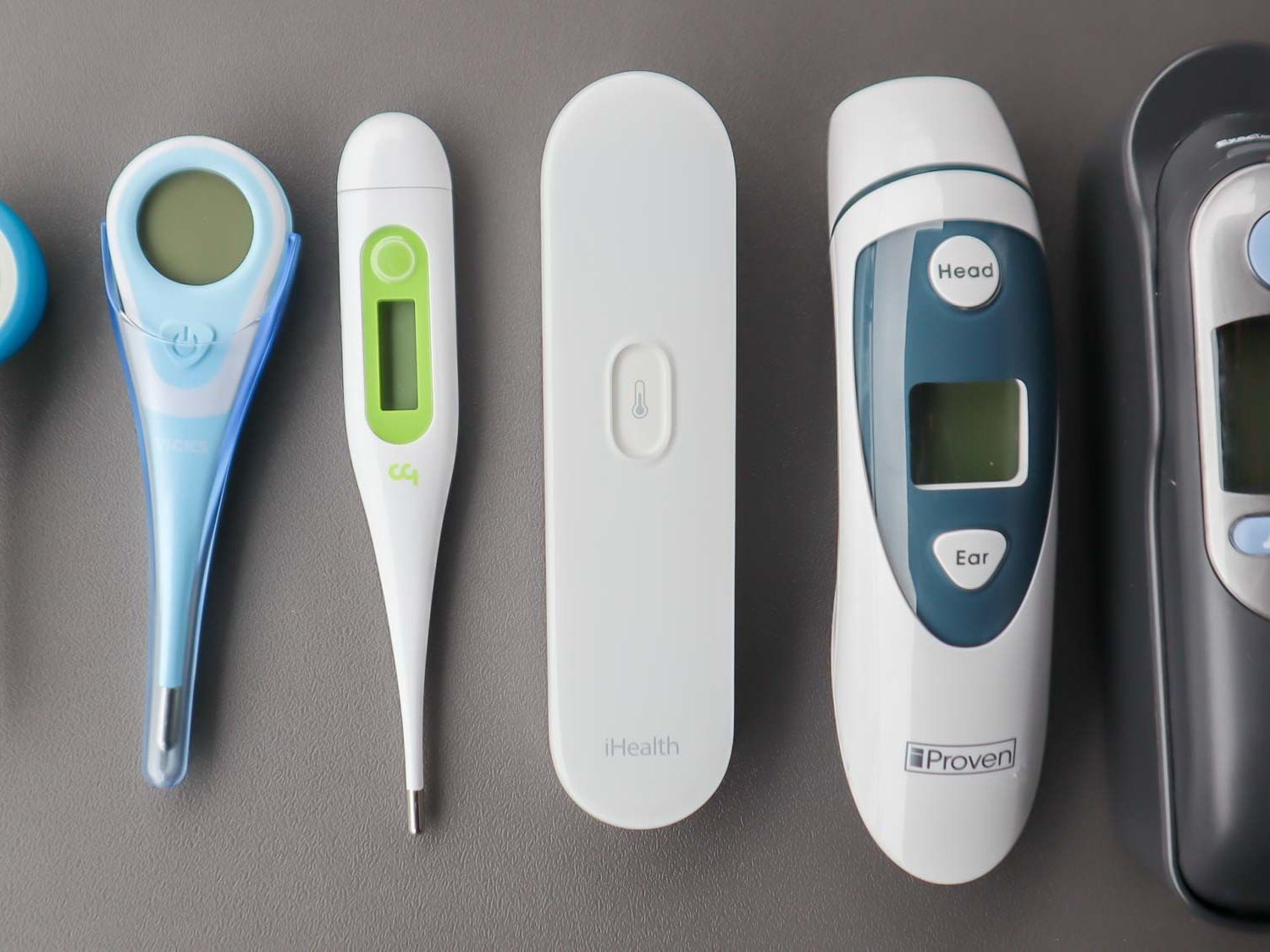
Understanding the relative accuracy of different thermometer types across pediatric age groups is essential for making informed clinical decisions.
Age-Specific Accuracy Recommendations
Infants (0-3 months):
- Most Accurate: Rectal digital thermometer
- Alternative: Temporal artery thermometer (validated models)
- Screening Only: Axillary digital thermometer
Toddlers (3 months-3 years):
- Most Accurate: Rectal digital thermometer
- Highly Accurate: Temporal artery thermometer
- Good Accuracy: Tympanic thermometer (6+ months)
Children (4+ years):
- Most Accurate: Oral digital thermometer
- Highly Accurate: Temporal artery thermometer
- Good Accuracy: Tympanic thermometer
Clinical Accuracy Standards
Fever Thresholds by Method:
- Rectal: ≥38.0°C (100.4°F)
- Oral: ≥37.6°C (99.7°F)
- Temporal Artery: ≥37.6°C (99.7°F)
- Tympanic: ≥37.6°C (99.7°F)
- Axillary: ≥37.4°C (99.3°F)
Infection Control and Safety Considerations
Infection control is paramount in pediatric clinics, making thermometer selection and maintenance critical for patient safety.
Disposable vs. Reusable Components
Disposable Elements:
- Probe covers for tympanic thermometers
- Single-use thermometers for high-risk patients
- Disposable sheaths for digital thermometers
Cleaning and Disinfection Protocols:
- Alcohol-based disinfectants for surface cleaning
- UV sanitization systems for advanced disinfection
- Dedicated thermometers for isolation patients
- Regular calibration checks to maintain accuracy
Safety Features
Child Safety Elements:
- Flexible tips to prevent injury
- Auto-shutoff functions to prevent overheating
- Non-toxic materials for all patient-contact surfaces
- Rounded edges to prevent cuts or scratches
Cost-Effectiveness Analysis for Pediatric Clinics

Selecting appropriate thermometers for pediatric clinics requires balancing accuracy, functionality, and cost-effectiveness.
Initial Investment Considerations
Budget Categories:
- Basic Digital Thermometers: $10-25 per unit
- Infrared Forehead Thermometers: $30-100 per unit
- Professional Tympanic Thermometers: $100-300 per unit
- Temporal Artery Systems: $200-500 per unit
Long-term Cost Analysis:
- Disposable supplies (probe covers, batteries)
- Maintenance and calibration requirements
- Replacement schedules based on usage volume
- Staff training and efficiency gains
Return on Investment Factors
Efficiency Gains:
- Reduced measurement time increases patient throughput
- Improved accuracy reduces repeat measurements
- Enhanced patient satisfaction through comfort and speed
- Decreased infection risk with non-contact methods
Best Practices for Thermometer Selection in Pediatric Clinics

Implementing an effective temperature measurement program requires strategic planning and systematic approach.
Selection Criteria Framework
Primary Considerations:
- Patient population demographics (age distribution)
- Clinical volume and throughput requirements
- Accuracy standards for specific patient conditions
- Budget constraints for initial and ongoing costs
- Staff training and competency requirements
Secondary Factors:
- Manufacturer reputation and clinical validation
- Technical support and warranty coverage
- Integration capabilities with existing systems
- Regulatory compliance and FDA clearance
Implementation Strategy
Phase 1: Assessment and Planning
- Current state analysis of existing equipment
- Staff surveys on preferences and challenges
- Patient feedback on comfort and experience
- Clinical outcome review for accuracy assessment
Phase 2: Pilot Testing
- Limited deployment of candidate thermometers
- Comparative accuracy studies against reference standards
- Staff training and competency assessment
- Patient satisfaction evaluation
Phase 3: Full Implementation
- Gradual rollout across clinic departments
- Comprehensive staff training programs
- Quality assurance monitoring
- Continuous improvement based on feedback
Training and Competency Requirements
Ensuring accurate temperature measurement requires comprehensive training programs for clinical staff.
Core Competencies
Technical Skills:
- Proper thermometer positioning for each device type
- Patient preparation techniques for optimal accuracy
- Troubleshooting common measurement issues
- Maintenance and calibration procedures
Clinical Judgment:
- Age-appropriate method selection based on patient condition
- Result interpretation and clinical correlation
- When to repeat measurements for verification
- Documentation requirements and standards
Ongoing Education
Regular Training Updates:
- New technology introductions and updates
- Best practice refinements based on clinical evidence
- Competency assessments and skill validation
- Quality improvement initiatives and feedback
Future Trends in Pediatric Thermometry
The field of pediatric temperature measurement continues to evolve with advancing technology and clinical research.
Emerging Technologies
Advanced Sensor Technology:
- Multi-sensor arrays for enhanced accuracy
- AI-powered measurement optimization
- Continuous monitoring capabilities
- Wireless connectivity for seamless data integration
Patient Experience Enhancements:
- Gamification elements to reduce anxiety
- Virtual reality distractions during measurement
- Voice-guided instructions for older children
- Personalized measurement protocols based on patient history
Clinical Research Developments
Ongoing Studies:
- Comparative accuracy across diverse populations
- Age-specific calibration improvements
- Environmental factor impact assessments
- Long-term outcome correlations
Conclusion: Optimizing Temperature Measurement in Pediatric Care
Selecting the best thermometers for pediatric clinics requires careful consideration of multiple factors including patient age demographics, clinical requirements, accuracy standards, and cost-effectiveness. The evidence clearly demonstrates that different thermometer types excel in specific pediatric applications:
For Infants (0-6 months): Rectal digital thermometers remain the gold standard for accuracy, with temporal artery thermometers providing an excellent non-invasive alternative for routine screening.
For Toddlers (6 months-3 years): Temporal artery and tympanic thermometers offer the best balance of accuracy, speed, and patient comfort, making them ideal for this challenging age group.
For Children (4+ years): Oral digital thermometers provide excellent accuracy when cooperation is possible, with temporal artery thermometers serving as versatile alternatives for all situations.
For High-Volume Clinics: Non-contact infrared thermometers optimize workflow efficiency while maintaining clinical accuracy, making them particularly valuable in busy pediatric practices.
The investment in appropriate pediatric thermometry equipment pays dividends through improved patient satisfaction, enhanced clinical accuracy, and increased operational efficiency. By carefully selecting age-appropriate thermometers and implementing comprehensive training programs, pediatric clinics can ensure they are providing the highest quality temperature measurement services to their young patients.
As technology continues to advance, the future of pediatric thermometry promises even greater accuracy, enhanced patient comfort, and seamless integration with electronic health records. Healthcare providers who stay current with these developments and maintain evidence-based selection criteria will be best positioned to deliver optimal pediatric care while maximizing their investment in temperature measurement technology.
The key to success lies in understanding that pediatric thermometry is not a one-size-fits-all solution, but rather requires a thoughtful, age-appropriate approach that prioritizes both clinical accuracy and patient comfort. By following the guidelines and recommendations outlined in this comprehensive guide, pediatric clinics can make informed decisions that benefit both their patients and their practice operations.























:max_bytes(150000):strip_icc()/GettyImages-649745556-59b57f4f845b3400100a8135.jpg)